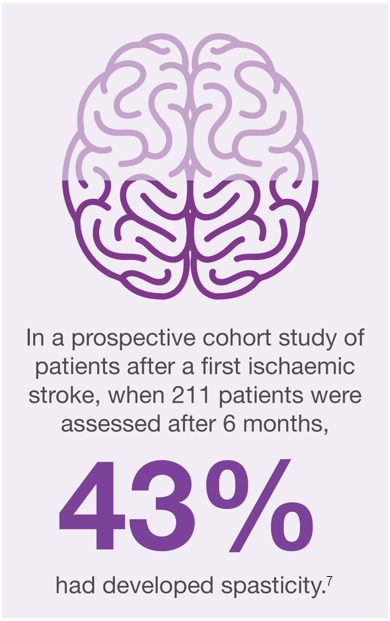
Post-stroke spasticity (PSS) has a significant impact on daily life6,7
PSS has a significant impact on a stroke survivor’s daily life, impeding basic tasks, such as eating and self-care6,8,9
PSS: post-stroke spasticity.
By clicking the link above you will leave the AbbVie Pro website and be taken to the eMC PI portal website.
Adverse events should be reported. Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/ or via the MHRA Yellow Card app, available in the Google Play or Apple App Stores.
Adverse events should also be reported to AbbVie on GBPV@abbvie.com
Date of preparation: June 2025. UK-BTX-250070.