BOTOX® has demonstrated efficacy in post‑stroke spasticity (PSS) in both the upper and lower limb5-9
BOTOX® has demonstrated efficacy in upper limb PSS6
BOTOX® helped significantly more patients achieve upper limb functional goals vs placebo6
Primary endpoint: Functional disability was measured using the four-point Disability Assessment Scale (hygiene, dressing, pain and limb position) at week 66
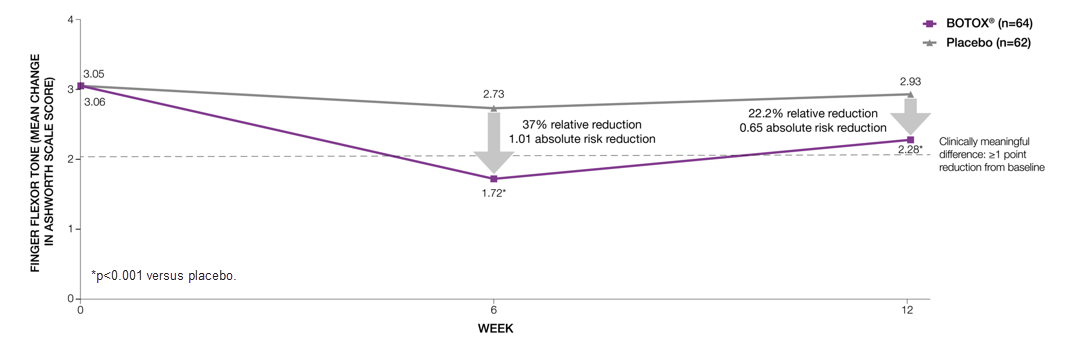
BOTOX® significantly reduced finger flexor tone compared to placebo at both Week 6 (primary endpoint) and 12 (additional endpoint)6
Adapted from Brashear A, et al 2002.6
Study context: A phase 3, randomised, double-blind, placebo-controlled, multicentre trial assessed the efficacy and safety of BOTOX® in 126 subjects with increased flexor tone in the wrist and fingers after a stroke6.
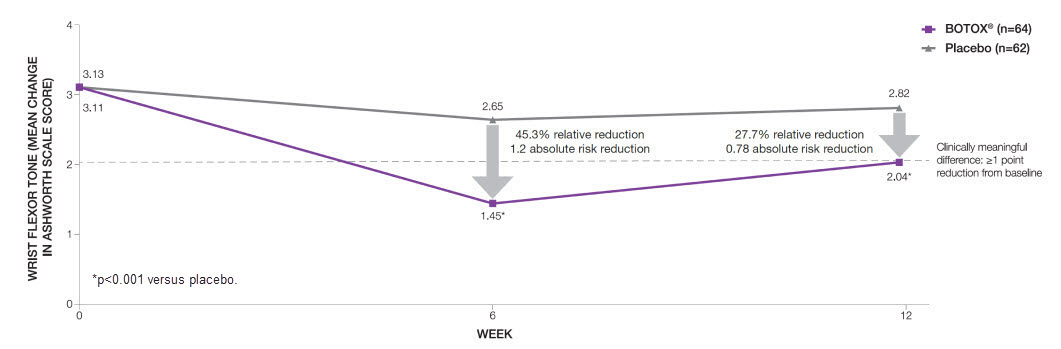
BOTOX® significantly* reduced wrist flexor tone compared to placebo at both Week 6 (primary endpoint) and 12 (additional endpoint)6
Adapted from Brashear A, et al 2002.6
Study context: A phase 3, randomised, double-blind, placebo-controlled, multicentre trial assessed the efficacy and safety of BOTOX® in 126 subjects with increased flexor tone in the wrist and fingers after a stroke.6
BOTOX® has demonstrated efficacy in lower limb PSS7-9
Primary endpoint: change from baseline in MAS for the ankle plantar flexors (average of Weeks 4 and 6)8
BOTOX® met the primary endpoint with improved mean change in ankle MAS at Weeks 4 to 6 from baseline vs placebo (P=0.01)8
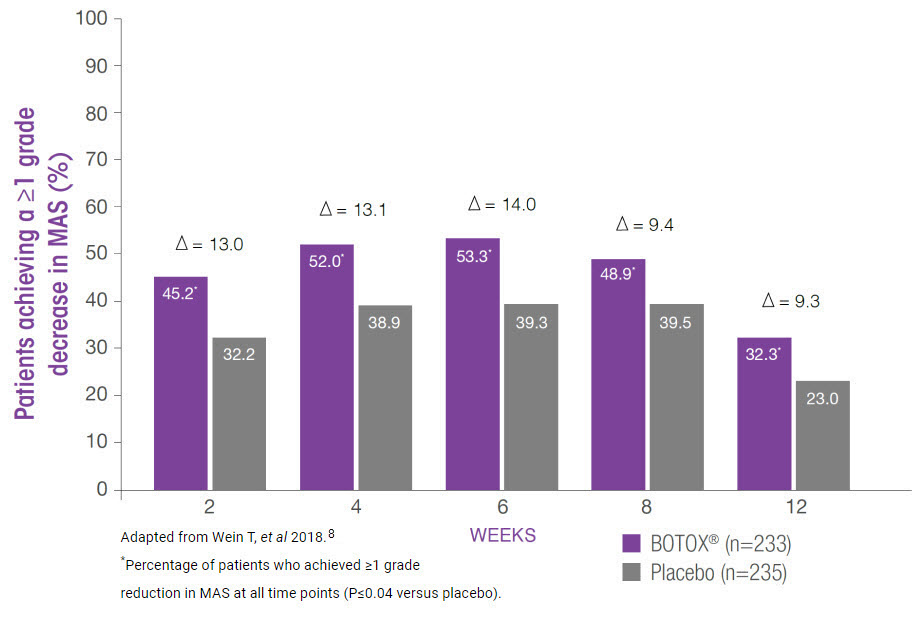
BOTOX® significantly improved ankle MAS scores by ≥1 grade vs placebo at all time points (additional endpoint)8
- The number of patients achieving a ≥1 grade decrease in ankle MAS continued to increase with repeated BOTOX® treatments during a subsequent 1-year open-label phase (additional endpoint)8
Study context: A phase 3, multicentre, randomised, double-blind, placebo-controlled trial (n=468, 12 weeks) with open-label extension (n=447, up to 60 weeks) evaluating the efficacy, safety and sustained benefit of BOTOX® in adults with post-stroke lower limb spasticity.8
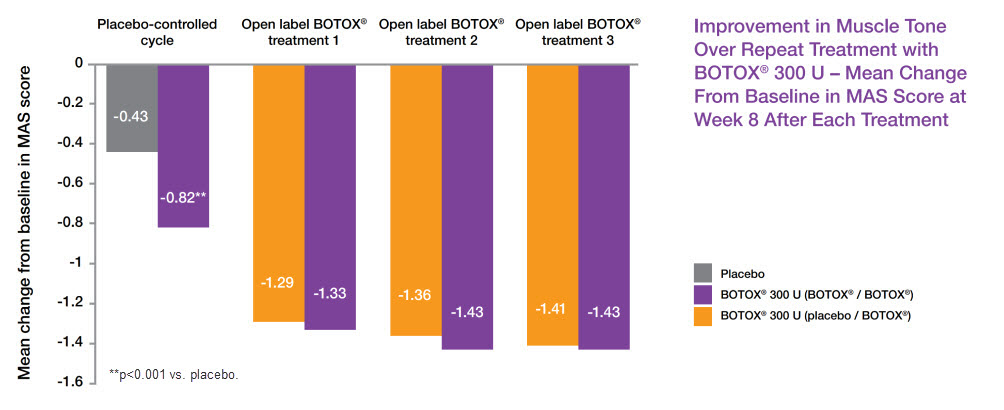
BOTOX®: Improvement in ankle muscle tone continued after repeated treatment (pre-specified secondary endpoint)7,9
Adapted from Allergan Data on File9
Study context: A phase 3, randomised, double-blind, multicentre, placebo-controlled study (n=120, 12 weeks), followed by open-label extension (n=112, up to 36 weeks), evaluating efficacy and safety of BOTOX® in patients with equinus deformity associated with PSLLS (BTX108512). Primary efficacy endpoint: AUC of change from baseline MAS ankle score during 12-week double-blind phase. Mean baseline of MAS ankle score: BOTOX® 3.28, Placebo 3.24.7,9
AUC, area under curve; MAS, Modified Ashworth Scale; PSLLS, post-stroke lower limb spasticity; PSS, post-stroke spasticity.
By clicking the link above you will leave the AbbVie Pro website and be taken to the eMC PI portal website.
Adverse events should be reported. Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/ or via the MHRA Yellow Card app, available in the Google Play or Apple App Stores.
Adverse events should also be reported to AbbVie on GBPV@abbvie.com
Date of preparation: June 2025. UK-BTX-250065.