This promotional material is intended for UK Healthcare Professionals only.
BOTOX® (botulinum toxin type A) Prescribing Information and adverse event reporting information can be found below.
The Patient Experience: Overactive bladder (OAB) can impact on patients' quality of life in many ways
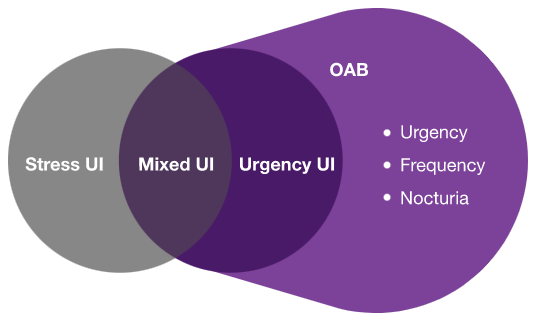
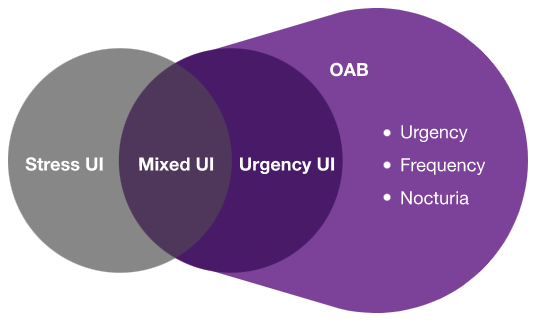
Definition of OAB: OAB is characterised by urinary urgency, with or without urgency urinary incontinence, usually with increased daytime frequency and nocturia6,7
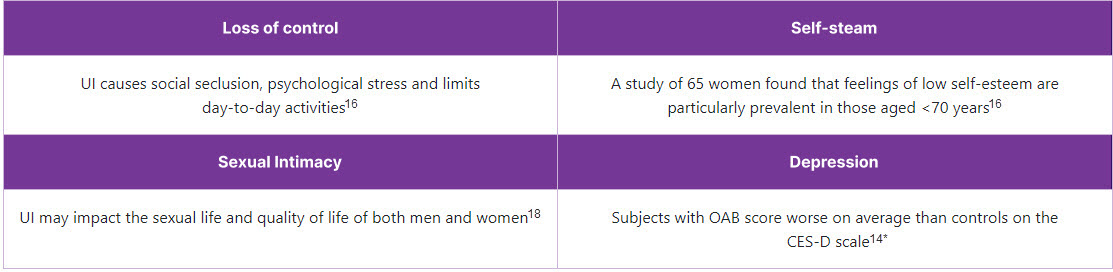
The impacts of OAB on patient quality of life are manifold8-16
Impact of OAB on daily living
2 out of 3 patients report that their symptoms affect their daily life17
*CES-D: Center for Epidemiological Studies – Depression. The CES-D is a self-reported depression scale including 20 items, developed to identify depression-related symptoms. Scores range from 0 to 60, with higher scores relating to more depression-related symptoms, and scores of 16 or over indicating depression.14
CES-D: Center for Epidemiological Studies - Depression; OAB: overactive bladder; UI: urinary incontinence.
Please refer to the BOTOX® Summary of Product Characteristics for further information on adverse events, contraindications and special warnings and precautions for use. The BOTOX® Summary of Product Characteristics can be found here
By clicking the link above you will leave the AbbVie Pro website and be taken to the eMC PI portal website.
Adverse events should be reported. Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/
Adverse events should also be reported to AbbVie on GBPV@abbvie.com
Date of preparation: May 2025. UK-BUO-250033.