This promotional material is intended for UK Healthcare Professionals only.
BOTOX® (botulinum toxin type A) Prescribing Information and adverse event reporting information can be found below.
Administering BOTOX® for your overactive bladder (OAB) patients
BOTOX® provides approximately 6 months of symptom relief6
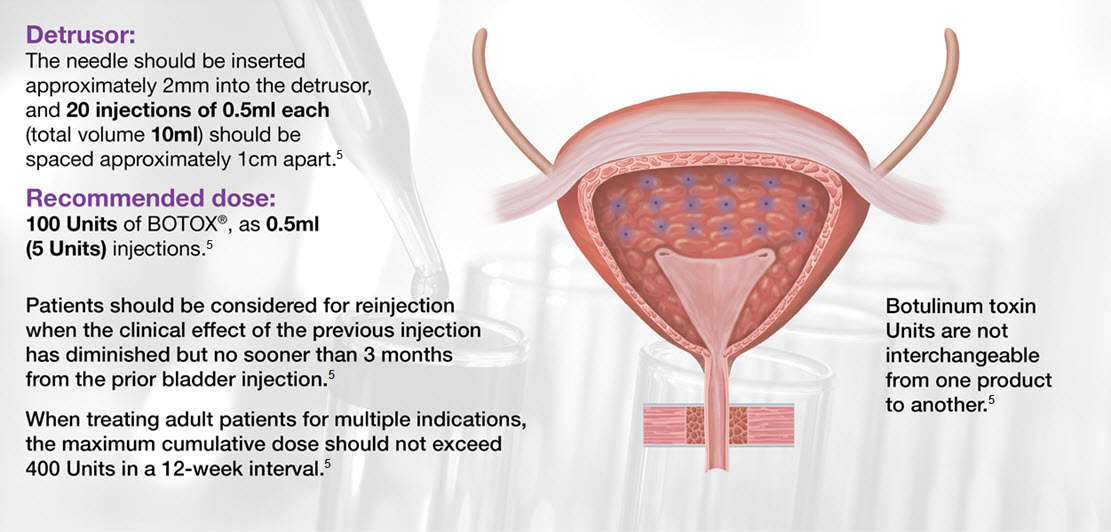
Please follow the SmPC for reconstitution protocol (section 6.6). For the patient preparation and post-injection monitoring, please see section 4.4 of the SmPC
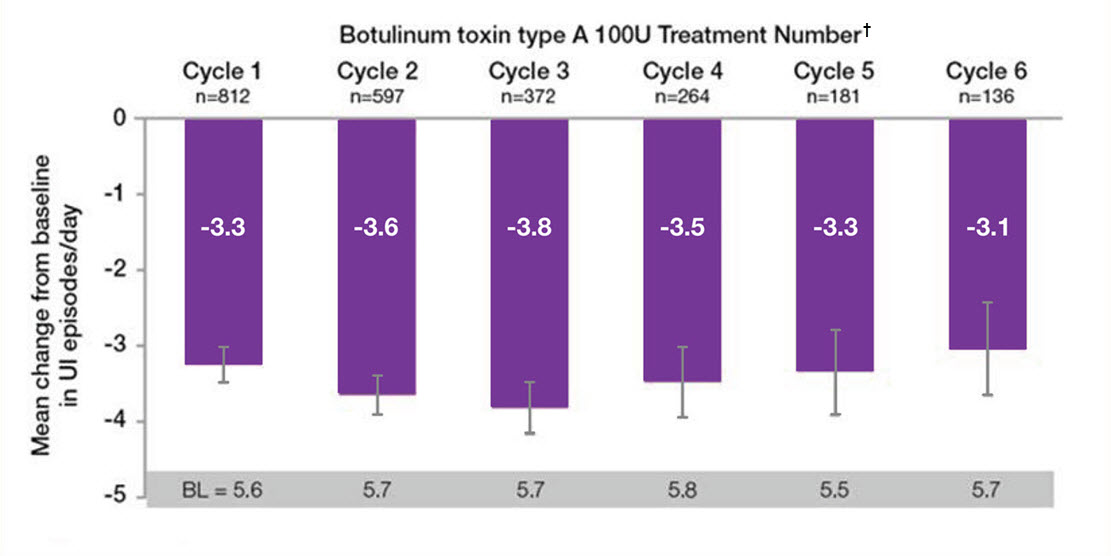
BOTOX® in overactive bladder: Treatment benefit sustained over time6
BOTOX® sustained reductions in daily episodes of UI and urgency from baseline, throughout the entire 3.5 year study period6
Adapted from Nitti VW, et al. 2016.6
Study context: The final results of the prospective, multicentre, long-term (3.5 year) study of the efficacy/safety of BOTOX® for overactive bladder syndrome. The co-primary endpoints were the mean reduction in UI episodes per day and the proportion of patients reporting a positive response on the treatment benefit scale. In the overall population the most common AEs within the first 12 weeks of BOTOX® treatment were localised to the urinary tract. There was no evidence of increasing occurence of these AEs with repeat treatments, UTI was the most frequently reported AE6
†n values denote the number of patients with data available at week 12 post treatment. Error bars represent 95% confidence intervals.
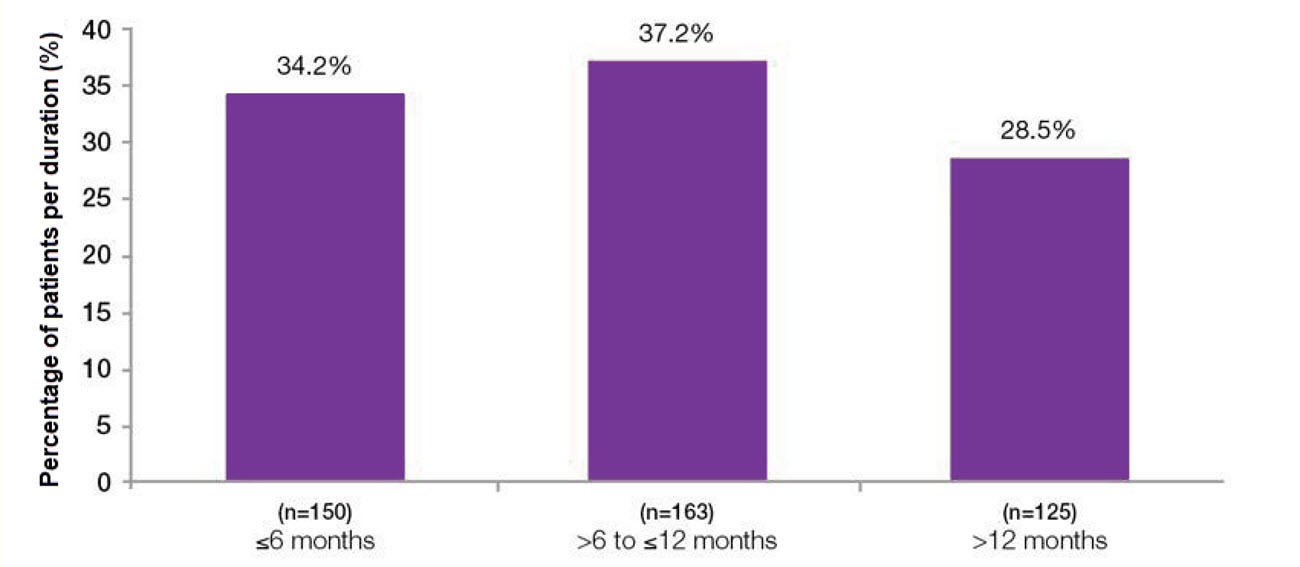
Overall median duration of effect was 7.6 months6
Duration of botulinum toxin type A effect based on time to patient request for re-treatment6
Adapted from Nitti VW, et al. 2016.6
Study context: The final results of the prospective, multicenter, long-term (3.5 year) study of the efficacy/safety of BOTOX® for overactive bladder syndrome. Median duration in patients (n=438) who received 100U BOTOX® only throughout study with complete treatment cycles, with 1 month defined as 4 weeks. Total study duration 3.5 years. Patients received up to 6 treatment cycles.6
AE: adverse event; BL: baseline; SmPC: summary of product characteristics; UI: urinary incontinence; UTI: urinary tract infection.
Please refer to the BOTOX® Summary of Product Characteristics for further information on adverse events, contraindications and special warnings and precautions for use. The BOTOX® Summary of Product Characteristics can be found here
By clicking the link above you will leave the AbbVie Pro website and be taken to the eMC PI portal website.
Adverse events should be reported. Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/
Adverse events should also be reported to AbbVie on GBPV@abbvie.com
Date of preparation: May 2025. UK-BUO-250038.











 Approximately 52% of patients completed the entire 3.5 year study6
Approximately 52% of patients completed the entire 3.5 year study6