Note to affiliates: This update to the venetoclax CLL AbbVie Pro site includes a homepage headline, updated CLL14 6-year, CLL13 4-year, MURANO 7-year data sets, and other streamlined content updates. CLL 13 4-year update reflects the CLL13 data from the Lancet Oncology publication. The CLL14 6-year and MURANO 7-year data have been updated based on the EHA 2023 abstracts. For countries that cannot use these data sets, please follow local regulations and MRLO guidance, and revert to CLL14 5-year and MURANO 5-year published data from the product label.
Primary analysis in ITT population for VEN+O vs O+Clb1:
INV-assessed PFS†: Reduced risk of progression or death (HR=0.35; 95% CI: 0.23–0.53 [P<0.0001]).
| • | Median follow-up of 28 months |
Additional analyses:
6-year PFS estimate (INV-assessed)2‡: 53% vs 22% (HR=0.40; 95% CI: 0.31–0.52) after 5 years off treatment.
| • | Median PFS of 76.2 months with VEN+O vs 36.4 months with O+Clb |
INV-assessed complete remission (CR/CRi)1: 50% vs 23% (P<0.0001).
| • | ORR: 85% (95% CI: 79.2–89.2) vs 71% (95% CI: 64.8–77.2 [P=0.0007]) |
Primary analysis in ITT population for VEN+R vs BR1:
INV-assessed PFS†: Reduced risk of progression or death (HR=0.17; 95% CI: 0.11–0.25 [P<0.0001]).
| • | Median follow-up of 23.8 months |
Additional analyses:
7-year PFS estimate (INV-assessed)3‡: 23% (HR=0.23; 95% CI: 0.18–0.29) vs NE after ~5 years off treatment.
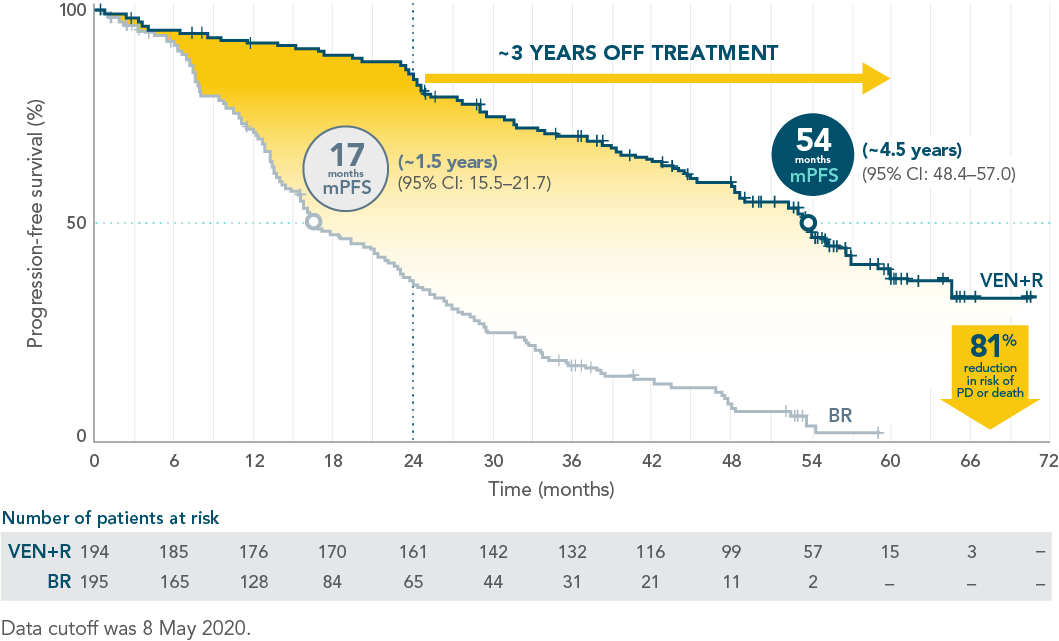
| • | Median PFS of 54.7 months with VEN+R (95% CI: 52.3–59.9) vs 17.0 months with BR (95% CI: 15.5–21.7) |
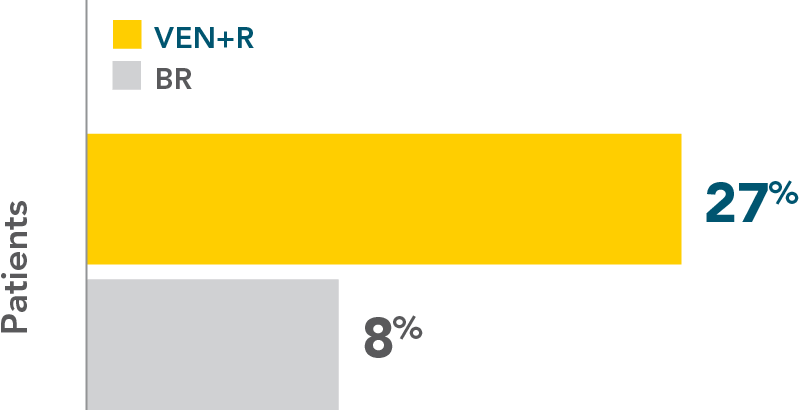
INV-assessed complete remission (CR/CRi)1‡: 27% vs 8%.
| • | ORR: 93% (95% CI: 88.8–96.4) vs 68% (95% CI: 60.6–74.2) |
*See full dosing information for VEN+O and for VEN+R in the dosing and administration section.
†Primary endpoint.
‡Results are descriptive only.
1L=first line; CLL=chronic lymphocytic leukaemia; VEN+O=VENCLYXTO + obinutuzumab; ITT=intent to treat; O+Clb=obinutuzumab + chlorambucil; INV=investigator; PFS=progression-free survival; HR=hazard ratio; CI=confidence interval; CR=complete remission; CRi=complete remission with incomplete bone marrow recovery; ORR=overall response rate; 2L+=second line + later lines of therapy; VEN+R=VENCLYXTO + rituximab; BR=bendamustine + rituximab; NE=not evaluable.
test
MURANO TRIAL WAS DESIGNED TO ALLOW PATIENTS TO COMPLETE VENCLYXTO + RITUXIMAB TREATMENT IN 2 YEARS1*
MURANO evaluated VENCLYXTO + rituximab vs a standard CIT regimen (bendamustine + rituximab)
*MURANO was a multicentre, randomised, open-label, phase 3 trial. Treatment complete after 5-week dose-titration period and twenty-four 28-day cycles.
†VENCLYXTO 400 mg daily after initial dose-titration period in the absence of disease progression or unacceptable toxicity.
‡Rituximab dosing: 375 mg/m2 IV Day 1, Cycle 1 (initiated after the 5-week dose titration schedule); 500 mg/m2 IV Cycles 2–6. Each cycle was 28 days.
§Bendamustine dosing: 70 mg/m2 IV Days 1 and 2, Cycles 1–6. Each cycle was 28 days.
||Venetoclax is taken for 24 months starting Day 1, Cycle 1 of rituximab.
Click here for titration protocols and full dosing information.
Select inclusion criteria2
| • | Previously treated with 1 to 3 prior therapies (including at least 1 chemotherapy-containing regimen) |
| • | Patients treated with prior bendamustine, provided the duration of response was ≥2 years |
Primary endpoint
| • | INV-assessed PFS¶ |
Select secondary endpoints
| • | IRC-assessed PFS |
| • | INV- and IRC-assessed ORR,# CR/CRi, nPR, and PR |
| • | Overall survival (OS) |
| • | TTNT |
| • | uMRD rates at EoCT** |
Primary analysis and results (ITT population)
Median follow-up of 23.8 months. Median PFS was not reached with VEN+R vs 17 months (95% CI: 15.5–21.6) with BR in the primary analysis.
| • | 2-year PFS estimate: 85% (95% CI: 79.1–90.6) with VEN+R vs 36% (95% CI: 28.5–44.0) with BR (HR=0.17; 95% CI: 0.11–0.25 [P<0.0001]) |
¶Assessed using the iwCLL updated NCI-WG guidelines (2008).
#ORR=CR/CRi+nPR+PR.
**MRD was evaluated in the PB and/or bone marrow using ASO-PCR and/or flow cytometry. The cutoff for an undetectable (negative) status was <1 CLL cell per 104 leukocytes.
CIT=chemoimmunotherapy; IV=intravenous; IRC=independent review committee; nPR=nodular partial remission; TTNT=time to next anti-leukaemic treatment; iwCLL=International Workshop on Chronic Lymphocytic Leukemia; NCI-WG=National Cancer Institute–sponsored Working Group.
VENCLYXTO + RITUXIMAB INDUCED DEEP RESPONSES,* INCLUDING HIGH RATES OF uMRD† IN PERIPHERAL BLOOD1
Unparalleled rates of uMRD at EoCT for VEN+R vs BR in 2L+ CLL
IN PERIPHERAL BLOOD
| • | In evaluable patients who completed 2 years of VEN+R treatment without progression, the rate of uMRD in PB at EoT was 64% in the VEN+R arm3,4 |
| • | Rates of uMRD in bone marrow at the EoCT were 16% (95% CI: 10.7–21.3) in the VEN+R arm and 1% (95% CI: 0.1–3.7) in the BR arm |
More than 3x as many patients achieved complete remission with VEN+R (27%) vs BR (8%)‡
COMPLETE REMISSION RATE (CR/CRi)
*Deep response as measured by uMRD or CR.
†uMRD (secondary endpoint) results in ITT population are presented. Results are descriptive. The cutoff for an undetectable (negative) status was <1 CLL cell per 104 leukocytes.
‡Results are descriptive.
Note to affiliates: This content below contains the 5-year MURANO PFS uMRD data from Kater J Clin Oncol 2020 supplementary appendix (see reference block for full citation). For regions that want to promote on the 2-year data, please reference Veeva job REF-50560 to access the 4-year analysis from ASH containing the 2-year EoT PFS uMRD data.
PFS WAS ASSESSED IN EVALUABLE PATIENTS WHO ACHIEVED uMRD IN PB AT EoT4*†
Median follow-up of 59 months (range 0–71.5 months).5
VEN+R: PFS RATES BASED ON MRD* STATUS IN PB AT EoT AT 5-YEAR ANALYSIS† (N=130)‡
*Data in evaluable patients who completed VEN+R without progressive disease. Not tested for statistical significance.1
†ASO-PCR and flow cytometry were used in the central analysis of serial PB MRD samples.4
‡uMRD=<10-4 CLL cell; L-MRD+=10-4 to <10-2 CLL cell; H-MRD+=≥10-2 CLL cell.4
§Missing/Other is defined as patients without MRD status at EoT.
Note to affiliates: This content below includes data from the current Summary of Product Characteristics.
LONGER LASTING PFS: 54 MONTHS mPFS FOR VENCLYXTO + RITUXIMAB VS 17 MONTHS FOR BR1
Updated 5-year analysis1*
HR=0.19 (95% CI: 0.15–0.26).
INV-ASSESSED PFS IN THE ITT POPULATION
*Results are descriptive.
mPFS=median progression-free survival; PD=progressive disease.
PFS benefit was sustained in patients off treatment for ~3 years
See MURANO study design section for primary analysis data.
Note to affiliates: The content below includes data from the current Summary of Product Characteristics.
VENCLYXTO + RITUXIMAB PFS BENEFIT WAS CONSISTENT ACROSS SUBGROUPS, INCLUDING HIGH-RISK PATIENTS1*†
Data cutoff: 8 May 2020.
PFS benefits in these high‑risk subgroups were sustained
beyond the 2 years of VEN+R treatment
*High-risk subgroups include Del(17p), TP53 deletion and/or mutation present, as well as IGHV unmutated.1
†Results are descriptive.
‡Unstratified hazard ratio is displayed on the x-axis with logarithmic scale.
TP53=tumour protein p53; IGHV=immunoglobulin heavy-chain variable gene; NE=not evaluable.
See SmPC for additional PFS subgroups.
Note to affiliates: This content below includes data taken from the current Summary of Product Characteristics. For those countries who cannot yet promote the 5-year OS analysis, please use 4-year OS data.
VENCLYXTO + RITUXIMAB REDUCED THE RISK OF DEATH COMPARED WITH BR1
In the primary OS analysis (ITT population; median follow-up 23.8 months), the HR for VEN+R vs BR was 0.48 (95% CI: 0.25–0.90). Median OS was not reached for either treatment group.
Updated 5-year OS analysis
HR=0.40 (95% CI: 0.26–0.62). Median OS was not reached for either treatment group.
UPDATED 5-YEAR ANALYSIS: OS IN THE ITT POPULATION*
*Results are descriptive.
OS=overall survival.
Note to affiliates: This content below includes data taken from the Seymour Blood 2022 article and its supplementary appendix (see reference block for full citation).
TIME TO NEXT TREATMENT FOR VENCLYXTO + RITUXIMAB PATIENTS AT 5-YEAR FOLLOW-UP5,6*
HR=0.26 (95% CI: 0.20–0.35).
MEDIAN TIME TO NEXT CLL TREATMENT
*Results are descriptive.
mTTNT=median time to next anti-leukaemic treatment.
Time off CLL treatment was sustained for 3 years in over half of 2L+ patients
Note to affiliates: This content below includes data taken from the Seymour Blood 2022 article and its supplementary appendix (see reference block for full citation).
RESULTS IN PATIENTS WHO RECEIVED SUBSEQUENT VENCLYXTO TREATMENT IN THE VEN+R ARM5,6*
| • | 67 VEN+R patients received subsequent CLL therapy after a median treatment-free interval of 23.7 months |
| • | Of these VEN+R patients, 32 were retreated with VENCLYXTO-based therapy |
| • | 18 of the 32 patients had an evaluable response. |
ORR TO SUBSEQUENT VEN-BASED THERAPY6
Median treatment duration: 11.4 months (range: 0.7–37.6)†
*Results are descriptive.
†Based on 2008 iwCLL guidelines for diagnosis, indications for treatment, response assessment, and supportive management of CLL.5
‡Out of 32 evaluable patients. Responses in patients who were treated with their next line of therapy for insufficient time to have their response assessed, or those patients who had no response assessments reported, were considered unevaluable.6
Best ORR=CR/CRi+PR/nPR.6
I want to find out
more
about VENCLYXTO
I want to receive more information about VENCLYXTO
References: 1. VENCLYXTO Summary of Product Characteristics. Ludwigshafen, Germany: AbbVie Deutschland GmbH & Co. KG. 2. Seymour JF, Kipps TJ, Eichhorst B, et al. Venetoclax–rituximab in relapsed or refractory chronic lymphocytic leukemia. N Engl J Med. 2018;378(12):1107-1120. 3. Kater AP, Wu JQ, Kipps T, et al. Venetoclax plus rituximab in relapsed chronic lymphocytic leukemia: 4-year results and evaluation of impact of genomic complexity and gene mutations from the MURANO phase III study. J Clin Oncol. 2020;38(34):4042-4054(suppl appendix). 4. Kater AP, Wu JQ, Kipps T, et al. Venetoclax plus rituximab in relapsed chronic lymphocytic leukemia: 4-year results and evaluation of impact of genomic complexity and gene mutations from the MURANO phase III study. J Clin Oncol. 2020;38(34):4042-4054. 5. Seymour JF, Kipps TJ, Eichhorst B, et al. Enduring undetectable MRD and updated outcomes in relapsed/refractory CLL after fixed-duration venetoclax-rituximab. Blood. 2022. https://doi.org/10.1182/blood.2021015014. Published online May 5, 2022. 6. Seymour JF, Kipps TJ, Eichhorst B, et al. Enduring undetectable MRD and updated outcomes in relapsed/refractory CLL after fixed-duration venetoclax-rituximab. Blood. 2022. https://doi.org/10.1182/blood.2021015014. Published online May 5, 2022. (suppl data).