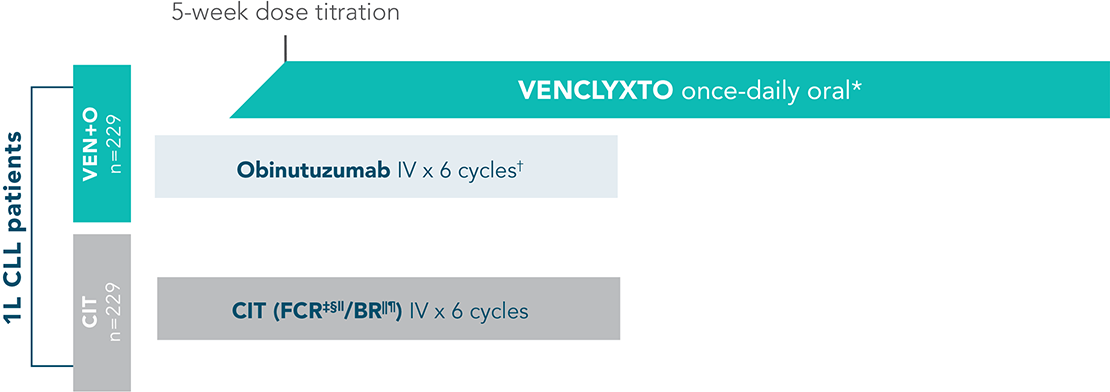Note to affiliates: This update to the venetoclax CLL AbbVie Pro site includes a homepage headline, updated CLL14 6-year, CLL13 4-year, MURANO 7-year data sets, and other streamlined content updates. CLL 13 4-year update reflects the CLL13 data from the Lancet Oncology publication. The CLL14 6-year and MURANO 7-year data have been updated based on the EHA 2023 abstracts. For countries that cannot use these data sets, please follow local regulations and MRLO guidance, and revert to CLL14 5-year and MURANO 5-year published data from the product label.
Primary analysis in ITT population for VEN+O vs O+Clb1:
INV-assessed PFS†: Reduced risk of progression or death (HR=0.35; 95% CI: 0.23–0.53 [P<0.0001]).
| • | Median follow-up of 28 months |
Additional analyses:
6-year PFS estimate (INV-assessed)2‡: 53% vs 22% (HR=0.40; 95% CI: 0.31–0.52) after 5 years off treatment.
| • | Median PFS of 76.2 months with VEN+O vs 36.4 months with O+Clb |
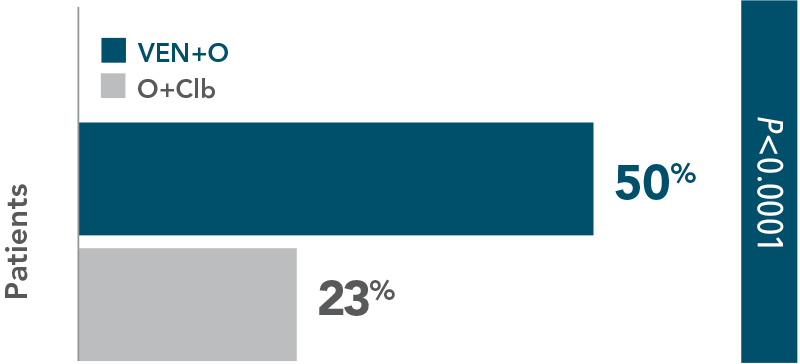
INV-assessed complete remission (CR/CRi)1: 50% vs 23% (P<0.0001).
| • | ORR: 85% (95% CI: 79.2–89.2) vs 71% (95% CI: 64.8–77.2 [P=0.0007]) |
Primary analysis in ITT population for VEN+R vs BR1:
INV-assessed PFS†: Reduced risk of progression or death (HR=0.17; 95% CI: 0.11–0.25 [P<0.0001]).
| • | Median follow-up of 23.8 months |
Additional analyses:
7-year PFS estimate (INV-assessed)3‡: 23% (HR=0.23; 95% CI: 0.18–0.29) vs NE after ~5 years off treatment.
| • | Median PFS of 54.7 months with VEN+R (95% CI: 52.3–59.9) vs 17.0 months with BR (95% CI: 15.5–21.7) |
INV-assessed complete remission (CR/CRi)1‡: 27% vs 8%.
| • | ORR: 93% (95% CI: 88.8–96.4) vs 68% (95% CI: 60.6–74.2) |
*See full dosing information for VEN+O and for VEN+R in the dosing and administration section.
†Primary endpoint.
‡Results are descriptive only.
1L=first line; CLL=chronic lymphocytic leukaemia; VEN+O=VENCLYXTO + obinutuzumab; ITT=intent to treat; O+Clb=obinutuzumab + chlorambucil; INV=investigator; PFS=progression-free survival; HR=hazard ratio; CI=confidence interval; CR=complete remission; CRi=complete remission with incomplete bone marrow recovery; ORR=overall response rate; 2L+=second line + later lines of therapy; VEN+R=VENCLYXTO + rituximab; BR=bendamustine + rituximab; NE=not evaluable.
test
CLL14 TRIAL WAS DESIGNED TO ALLOW PATIENTS TO COMPLETE VENCLYXTO + OBINUTUZUMAB TREATMENT IN 1 YEAR1*
CLL14 evaluated VENCLYXTO + obinutuzumab vs a standard CIT regimen (obinutuzumab + chlorambucil)
*CLL14 was a multicentre, randomised, open-label, phase 3 trial. Treatment complete after twelve 28-day cycles.
†VENCLYXTO 400 mg daily after initial dose-titration period.
‡Obinutuzumab dosing: 100 mg Cycle 1, Day 1, followed by 900 mg on Days 1 or 2; 1000 mg on Days 8 and 15 of Cycle 1 and Day 1 of Cycles 2–6.
§Chlorambucil dosing: 0.5 mg/kg on Days 1 and 15 of each 28-day cycle.
Click here for titration protocols and full dosing information.
Select inclusion criteria
| • | Previously untreated CLL and coexisting medical conditions (total CIRS >6 or CrCl <70 mL/min), age ≥18 years |
Select clinical endpoints||¶
| • | Primary endpoint: INV-assessed PFS |
| • | Select secondary endpoints: IRC-assessed PFS, ORR, CR/CRi, PR, TTNT, and uMRD# rate at end of treatment1,2 |
Primary analysis and results (ITT population)
| Median follow-up of 28 months (range: 0–36 months; VEN+O: n=216, O+Clb: n=216). Median PFS was not reached in either arm at primary analysis. |
| • | 2-year PFS estimate: 88% (95% CI: 83.7–92.6) with VEN+O vs 64% (95% CI: 57.4–70.8) with O+Clb (HR=0.35; 95% CI: 0.23–0.53 [P<0.0001]) |
||Assessed using the International Workshop for Chronic Lymphocytic Leukemia (iwCLL) updated National Cancer Institute–sponsored Working Group (NCI-WG) guidelines (2008).
¶ORR, CR/CRi, PR, and uMRD were assessed at EoT.
#MRD was evaluated using ASO-PCR. In PB or bone marrow, the cutoff for an undetectable (negative) status was <1 CLL cell per 104 leukocytes.
CIT=chemoimmunotherapy; IV=intravenous; CIRS=Cumulative Illness Rating Scale; CrCl=creatinine clearance; IRC=independent review committee; TTNT=time to next anti‑leukaemic treatment.
VENCLYXTO + OBINUTUZUMAB INDUCED DEEP RESPONSES,* INCLUDING uMRD, IN THE MAJORITY OF PATIENTS1
Unparalleled rates of uMRD for VEN+O vs O+Clb in 1L CLL
| • | Rates of uMRD in PB at EoT were 2 times higher in the VEN+O arm than in the O+Clb arm |
| • | Rates of uMRD in bone marrow at EoT were 3 times higher in the VEN+O arm (57% [95% CI: 50.1–63.6]) than in the O+CIb arm (17% [95% CI: 12.4–22.8]) (P<0.0001) |
uMRD (secondary endpoint) results in ITT population are presented. The cutoff for an undetectable (negative) status was <1 CLL cell per 104 leukocytes.
More than twice as many patients achieved complete remission with VEN+O (50%) vs O+Clb (23%)
COMPLETE REMISSION (CR/CRi) RATE†
| • | ORR‡ was 85% (95% CI: 79.2–89.2) in the VEN+O arm vs 71% (95% CI: 64.8–77.2) in the O+Clb arm (P=0.0007) |
*Deep response as measured by uMRD or CR.
†CR/CRi were assessed in the ITT population.2
‡ORR=CR/CRi+PR (secondary endpoint).
Note to affiliates: This content below contains 5-year PFS data from the Al-Sawaf HemaSphere 2022 article (see reference block for full citation). For countries that cannot yet promote this data, please use the 3-year follow-up data.
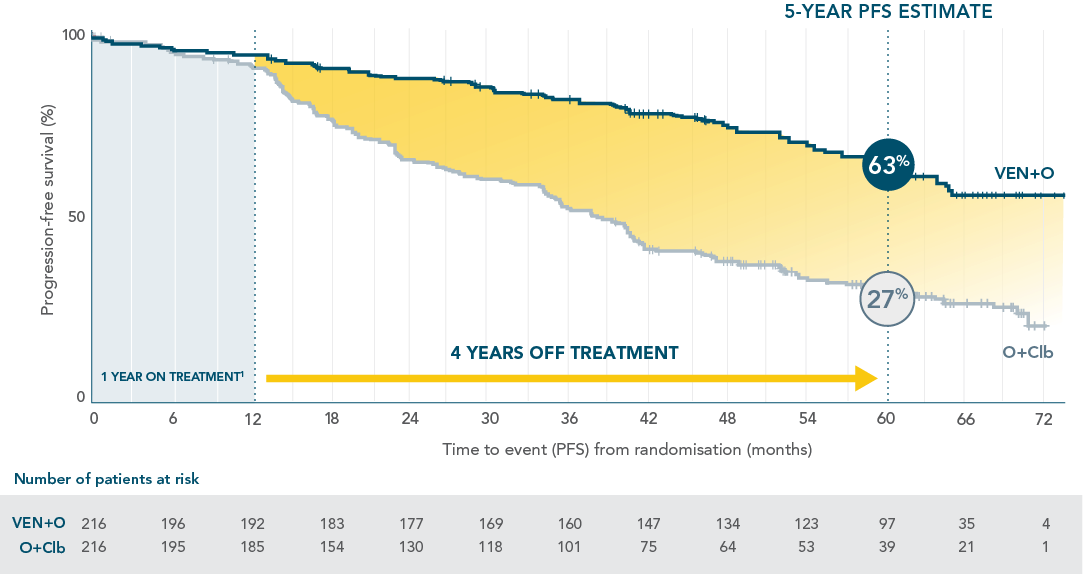
LONGER LASTING PFS: 63% PFS ESTIMATE FOR VENCLYXTO + OBINUTUZUMAB VS 27% FOR O+Clb AT 5 YEARS3*
Updated 5-year analysis
Median follow-up of 65.4 months (interquartile range: 52.6–69.4).
Median PFS was not reached with VEN+O vs 36.4 months with O+Clb (HR=0.35; 95% CI: 0.26–0.46).
INV-ASSESSED PFS IN THE ITT POPULATION
PFS benefit was sustained in patients off treatment for 4 years
*Results are descriptive.
Note to affiliate: the OPTIONAL content below is for regions with local regulations that do not allow 4-year or 5-year follow-up PFS data. This page has been created to emphasise 3-year follow-up data that appears in the “Al-Sawaf O, Zhang C, Tandon M, et al. Lancet Oncol 2020” publication and is not included in the SmPC.
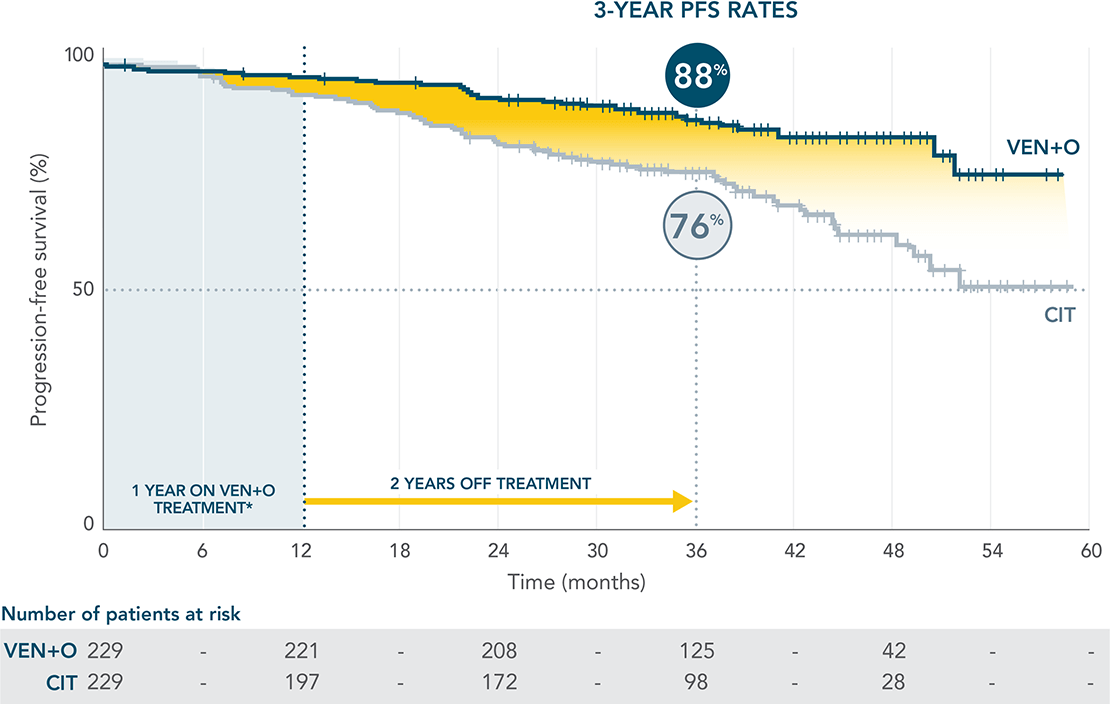
LONGER LASTING PFS: 82% PFS ESTIMATE FOR VENCLYXTO + OBINUTUZUMAB VS 50% FOR O+Clb AT 3 YEARSXX*
Updated 3-year analysis
Median follow-up of 40 months. Median PFS not reached with VEN+O; 35.6 months (95% CI: 33.7–40.7) with O+Clb
(HR=0.31; 95% CI: 0.22–0.44).
INV-ASSESSED PFS IN THE ITT POPULATION
Data cutoff was 23 August 2019.
PFS benefit was sustained in patients off treatment for 2 yearsXX
*Results are descriptive.
Reference: XX. Al-Sawaf O, Zhang C, Tandon M, et al. Venetoclax plus obinutuzumab versus chlorambucil plus obinutuzumab for previously untreated chronic lymphocytic leukaemia (CLL14): follow-up results from a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2020;21(9):1188-1200.
Note to affiliates: This content below contains 5-year TTNT data from the Al-Sawaf HemaSphere 2022 article (see reference block for full citation). For countries that cannot yet promote on this data, please use 4-year TTNT.
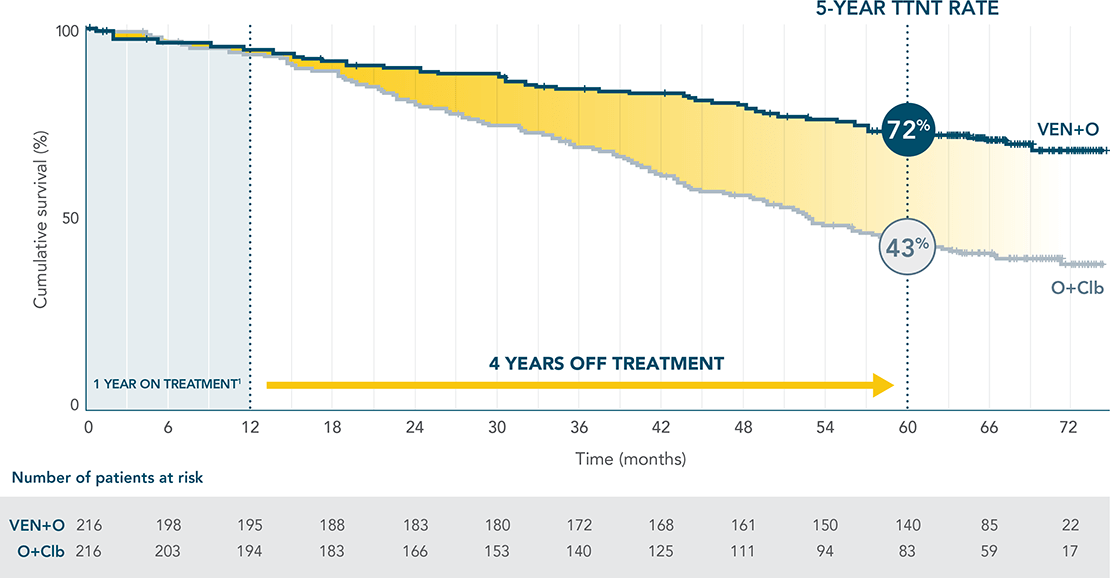
4 YEARS AFTER TREATMENT COMPLETION, 72% OF VENCLYXTO + OBINUTUZUMAB PATIENTS DID NOT REQUIRE NEXT TREATMENT3
Median TTNT was not reached with VEN+O vs 52.9 months with O+Clb (HR=0.42; 95% CI: 0.31–0.57).
Results are descriptive; not tested for statistical significance.
PERCENTAGE OF PATIENTS FREE OF NEXT CLL TREATMENT
TTNT=time to next anti-leukaemic treatment.
Note to affiliate: the OPTIONAL content below is for regions with local regulations that do not allow 5-year TTNT data. This page contains 4-year TTNT data from the Al-Sawaf J Clin Oncol 2021 publication and the Al-Sawaf publication in HemaSphere 2021.
3 YEARS AFTER TREATMENT COMPLETION, 81% OF VENCLYXTO + OBINUTUZUMAB PATIENTS DID NOT REQUIRE NEXT TREATMENTXX,YY
HR=0.46; 95% CI: 0.32–0.65 (P<0.0001).
PERCENTAGE OF PATIENTS FREE OF NEXT CLL TREATMENT*
The majority of patients who received 1 year of VEN+O remained treatment free at 4 years1
*Results are descriptive.
TTNT=time to next treatment.
References: XX. Al-Sawaf O, Zhang C, Robrecht S, et al. Venetoclax-obinutuzumab for previously untreated chronic lymphocytic leukemia: 4-year follow-up analysis of the randomized CLL14 study. HemaSphere. 2021;5:(S2):27. YY. Al-Sawaf O, Zhang C, Lu T, et al. Minimal residual disease dynamics after venetoclax-obinutuzumab treatment: extended off-treatment follow-up from the randomized CLL14 study. J Clin Oncol. 2021;39(36):4049-4060.
Note to affiliates: This content below includes data published in Al-Sawaf J Clin Oncol 2021 (see reference block for full citation).
VENCLYXTO + OBINUTUZUMAB PFS BENEFIT WAS CONSISTENT ACROSS HIGH-RISK SUBGROUPS AT 4-YEAR FOLLOW-UP4*
4-year analysis
Median follow-up of 52 months (interquartile range: 49.5–56.2). Median PFS was not reached for the VEN+O arm vs 36.4 months in the O+Clb arm.
| • | 4-year PFS estimate: 74% with VEN+O vs 35% with O+Clb (HR=0.33; 95% CI: 0.25–0.45) |
INV-ASSESSED PFS BY SUBGROUP AT 4 YEARS5
Data cutoff: 11 September 2020.4
*High-risk subgroups include Del(17p), TP53 deletion and/or mutation present, as well as IGHV unmutated.4
†Results are descriptive.
TP53=tumour protein p53; IGHV=immunoglobulin heavy-chain variable gene.
According to global Regulatory guidance, if this CLL13 content is incorporated into the Web site, the Deep Responses V-Chart in section 1.0 must be removed, reference numbers in the remaining sections must be revised, and references for the page must be removed from the reference block.
UPDATED PHASE 3 STUDY RESULTS FROM CLL13 IN 1L CLL6-8
VENCLYXTO + obinutuzumab vs CIT (FCR/BR) arms
CLL13 was a multicentre, open-label, phase 3, four-arm trial of treatment-naive, fit (CIRS ≤6, normal creatinine clearance ≥70 mL/min) patients with CLL. Patients were randomised to receive CLL therapy, including CIT (FCR for patients ≤65 years; BR for patients >65 years) or a VENCLYXTO regimen, including VENCLYXTO + obinutuzumab (further details for the study NCT02950051 are available at clinicaltrials.gov). Only those arms on-label for VENCLYXTO are detailed below.
DOSING SCHEMA FOR VENCLYXTO + OBINUTUZUMAB AND CIT ARMS
*VENCLYXTO 400 mg daily after initial dose-titration period for 12 cycles. Each cycle was 28 days.
†Obinutuzumab dosing: 1000 mg on Days 1, 8, and 15 of Cycle 1 and Day 1 of Cycles 2–6. Each cycle was 28 days.
‡Fludarabine dosing: 25 mg/m2 on Days 1–3 of Cycles 1–6. Each cycle was 28 days.
§Cyclophosphamide dosing: 250 mg/m2 on Days 1–3 of Cycles 1–6. Each cycle was 28 days.
IIRituximab dosing: 375 mg/m2 on Day 1, Cycle 1, and 500 mg/m2 on Day 1, Cycles 2–6. Each cycle was 28 days.
¶Bendamustine dosing: 90 mg/m2 on Days 1–2 of Cycles 1–6. Each cycle was 28 days.
Co-primary endpoints
| • | uMRD in peripheral blood at Month 15 assessed by flow cytometry (VEN+O vs CIT). uMRD was defined as <1 CLL cell per 104 leukocytes |
| • | PFS (for unapproved regimen not shown) |
Select secondary endpoints
| • | PFS (VEN+O vs CIT) |
| • | ORR# and CR/CRi# |
| • | Bone marrow uMRD was evaluated 2 months after end of treatment in patients with clinical CR |
Select inclusion criteria
| • | Creatinine clearance ≥70 mL/min |
Select exclusion criteria
| • | del(17p) or TP53 mutation |
#Assessed at Month 15 according to iwCLL 2008 guidelines.
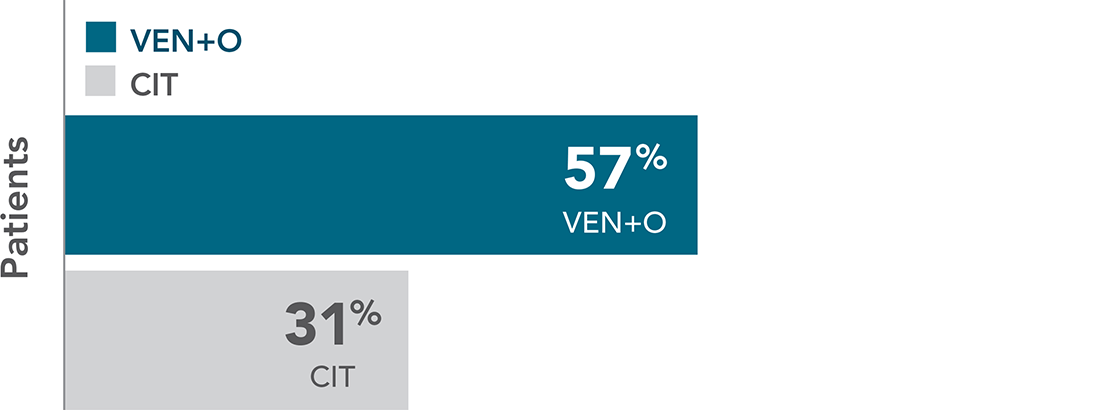
87% OF VENCLYXTO + OBINUTUZUMAB-TREATED PATIENTS ACHIEVED uMRD IN PERIPHERAL BLOOD VS 52% FOR CIT7
The median observation time was 27.9 months.
IN PERIPHERAL BLOOD
| • | Rates of uMRD in bone marrow were 73% in the VEN+O arm vs 37% in the CIT arm |
RATES OF CR/CRi IN VEN+O VS CIT
| • | INV-assessed ORR (CR/CRi+PR) was 96% in the VEN+O arm vs 81% in the CIT arm |
VENCLYXTO + OBINUTUZUMAB PFS RESULTS IN 1L FIT CLL7-9
A pre-planned interim analysis of PFS was conducted at Month 61. At the cutoff date (20 January 2022), the median observation time was 38.8 months. Because the PFS co-primary endpoint includes an unapproved regimen, these results are not shown.
Secondary endpoint: PFS for VEN+O vs CIT
Median follow-up of 38.8 months. (HR=0.42; 97.5% CI: 0.26–0.68, P<0.0001).
INV-ASSESSED PFS ANALYSIS
SAFETY INFORMATION FROM THE CLL13 TRIAL
| • | In CLL13, the most common Grade 3–5 treatment-emergent AEs were neutropaenia (51% of all patients), thrombocytopaenia (12%), TLS (8%), infusion-related reaction (7%), febrile neutropaenia (7%), and pneumonia (5%). There were no fatal cases of TLS and no patients required dialysis due to TLS |
| • | For the VEN+O and CIT arms, secondary neoplasia occurred in 23 and 36 patients, respectively. Incidence of CTC Grade 3–4 infections was 13.2% and 18.5%, respectively. Fatal AEs occurred in 9 and 10 of the VEN+O- and CIT-treated patients, respectively |
*CIT was given for a total of 6 cycles.
FCR=fludarabine + cyclophosphamide + rituximab; AE=adverse event; TLS=tumour lysis syndrome; CTC=common toxicity criteria.
I want to find out
more
about VENCLYXTO
I want to receive more information about VENCLYXTO
References: 1. VENCLYXTO Summary of Product Characteristics. Ludwigshafen, Germany: AbbVie Deutschland GmbH & Co. KG. 2. Fischer K, Al-Sawaf O, Baho J, et al. Venetoclax and obinutuzumab in patients with CLL and coexisting conditions. N Engl J Med. 2019;380(23):2225-2236. 3. Al-Sawaf O, Zhang C, Robrecht S, et al. Venetoclax-obinutuzumab for previously untreated chronic lymphocytic leukemia: 5-year results of the randomized CLL14 study. HemaSphere. 2022;6:(S3):49-50. 4. Al-Sawaf O, Zhang C, Lu T, et al. Minimal residual disease dynamics after venetoclax-obinutuzumab treatment: extended off-treatment follow-up from the randomized CLL14 study. J Clin Oncol. 2021;39(36):4049-4060. 5. Al-Sawaf O, Zhang C, Tandon M, et al. Minimal residual disease dynamics after venetoclax-obinutuzumab treatment: extended off-treatment follow-up from the randomized CLL14 study. J Clin Oncol. 2021;39(36):4049-4060(suppl appendix). 6. NCT02950051. ClinicalTrials.gov. Published October 31, 2016. Updated September 30, 2021. Accessed December 8, 2021. https://clinicaltrials.gov/ct2/show/NCT02950051? term=NCT02950051&draw=2&rank=1 7. Eichhorst B, Niemann C, Kater AP, et al. Blood. 2021;138(Supplement 1):71. 8. Eichhorst B, Niemann CU, Kater AP, et al. HemaSphere. 2022;6:S3. 9. Fürstenau M, Robrecht S, Zhang C, et al. Blood. 2021;138(Supplement 1):2639.