SIMPLICITY
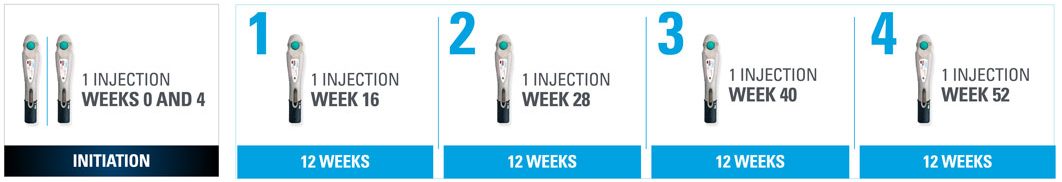
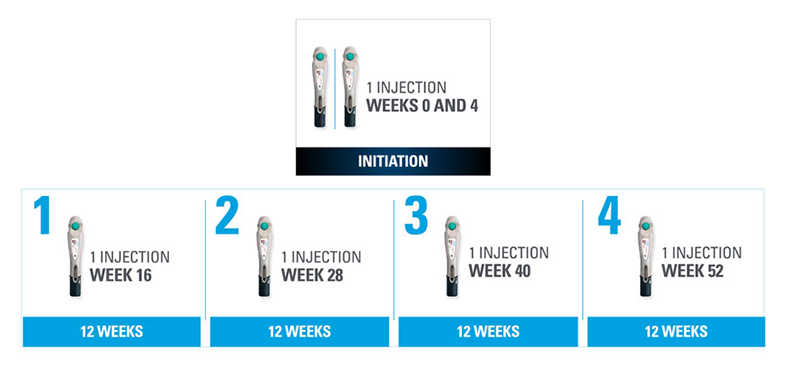
Nothing more than 4 INJECTIONS PER YEAR after initiation doses for both PsO and PsA patients*
- NO DOSE ADJUSTMENT required regardless of baseline characteristics, including BMI and weight1-4†
- SKYRIZI is dosed 150 mg (one 150-mg subcutaneous injection) at Week 0, Week 4, and every 12 weeks thereafter1
- 1 injection/dose for both the SKYRIZI prefilled pen and prefilled syringe
| * | Maintenance dosing (1 injection/dose) every 12 weeks following a starter dose at Week 0 and Week 4. |
| † | Risankizumab clearance and volume of distribution increase as body weight increases, which may result in reduced efficacy in subjects with high body weight (>130 kg). However, this observation is based on a limited number of subjects. |
SKYRIZI one injection per dose:
SAME EFFICACY AND SAFETY PROFILE
Same active ingredient | Demonstrated bioequivalence
NOW EVEN SIMPLER WITH
SKYRIZI 150 mg bioequivalence data1
Bioequivalence was demonstrated between a single SKYRIZI 150-mg injection and two SKYRIZI 75-mg injections in a prefilled syringe. Bioeqivalence was also demonstrated between SKYRIZI 150 mg in a prefilled syringe and a prefilled pen.
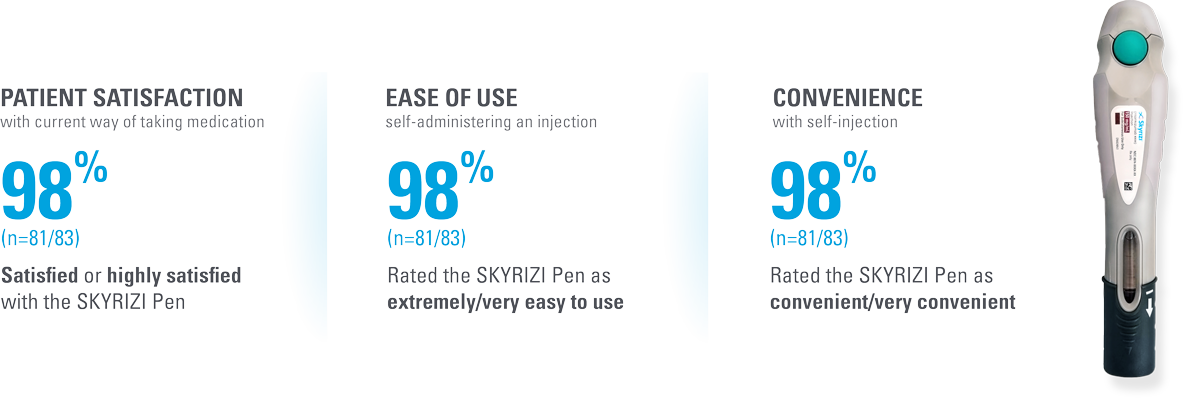
The SKYRIZI prefilled pen scores high across multiple measures of patient satisfaction6*†
| * | Patient satisfaction with self-injection was measured at Week 28 (4 injections total) in patients using the SKYRIZI Pen by the Self-Injection Assessment Questionnaire (SIAQ), a validated instrument for measuring patient feelings and experiences with self-injection. Score measures proportion of subjects achieving the best two categories in post-dose SIAQ by visit (observed cases; intention-to-treat population).6 |
| † | Patient satisfaction was also assessed for (n=83, observed cases): • Satisfied with frequency of injection (97.6%) • Satisfied with duration of injection (96.4%) • Continue self-injecting your medication (100.0%) • Continue self-injecting at home (97.6%) |
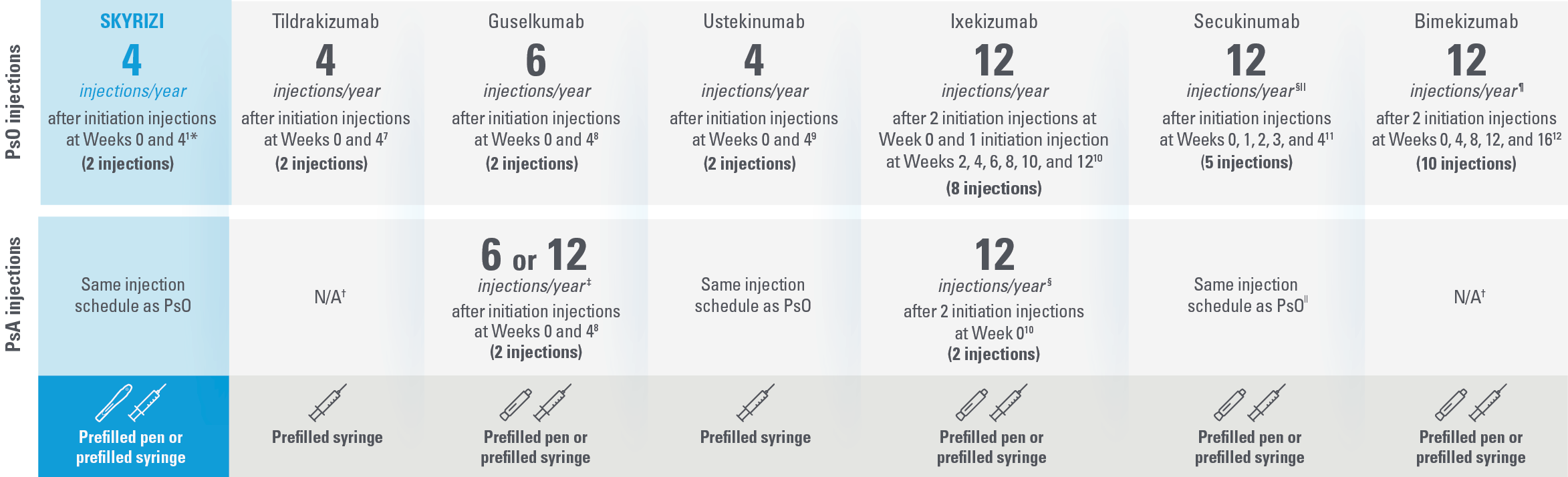
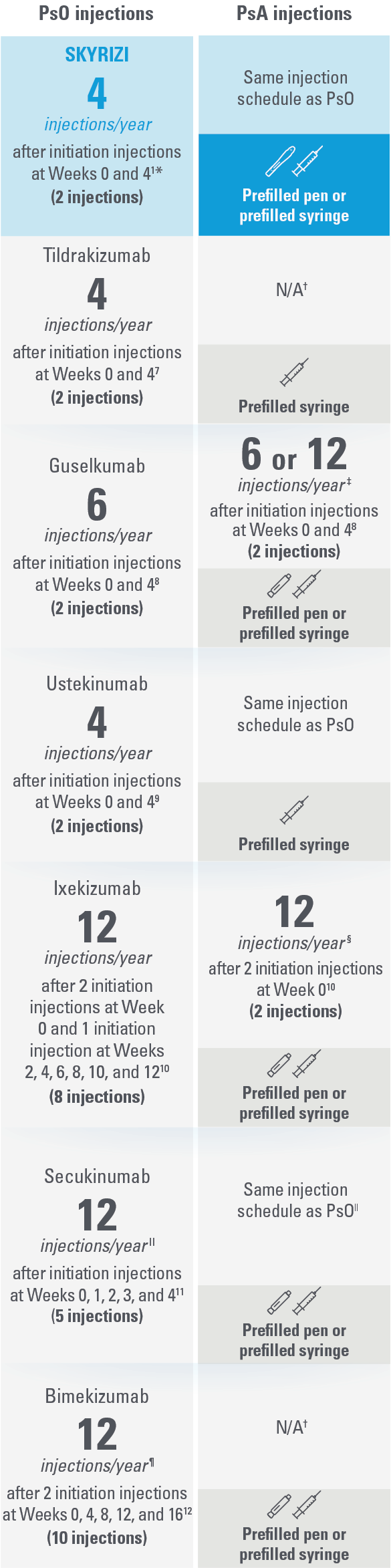
SKYRIZI: The ONLY treatment for adults with PsO and/or PsA with just 4 injections per year* and a prefilled pen1,7-12
This presentation is not intended to compare the efficacy or safety of the treatments shown. While these factors are important, there are additional considerations for selecting a treatment. Please refer to each productʼs Summary of Product Characteristics for additional information.
| The table above reflects devices available as of Januray 2022. | |
| * | Maintenance dosing (one 150-mg subcutaneous injection/dose) every 12 weeks following a starter dose at Week 0 and Week 4. |
| † | Not indicated for psoriatic arthritis as of January 2022.7,12 |
| ‡ | For PsA patients at high risk for joint damage according to clinical judgment, a dose of 100 mg every 4 weeks may be considered, instead of every 8 weeks.8 |
| § | The recommended dose is 160 mg by subcutaneous injection (two 80-mg injections) at Week 0, followed by 80 mg (one injection) every 4 weeks thereafter. For PsA patients with concomitant moderate to severe PsO, the recommended dosing regimen is the same as for PsO.10 |
| II | Patients with PsA with concomitant moderate to severe PsO or who are anti-TNFα inadequate responders, a 300-mg dose is recommended. Dose adjustment may be needed in certain patients depending on clinical response.11 |
| ¶ | For some patients with a body weight of ≥120 kg who did not achieve complete skin clearance at Week 16, dose recommendation is 2 injections every 4 weeks after Week 16 instead of every 8 weeks.12 |
SKYRIZI is intended for use under the guidance of a healthcare professional. Patients may self-inject SKYRIZI after training in subcutaneous injection technique. Instruct patients to inject the full 150-mg dose and to read the Instructions for Use before administration.
Find out more about SKYRIZI
References
- SKYRIZI [Summary of Product Characteristics]. AbbVie Ltd; November 2021.
- Leonardi C, Gordon K, Longcore M, Gu Y, Puig L. Weight-based analysis of psoriasis area and severity index improvement at 52 weeks of risankizumab or ustekinumab treatment: an integrated analysis of patients with moderate-to-severe plaque psoriasis. Presented at: 24th World Congress of Dermatology (WCD); June 10–15, 2019; Milan, Italy. Poster 5248.
- Strober B, Menter A, Leonardi C, et al. Efficacy of risankizumab in patients with moderate-to-severe plaque psoriasis by baseline demographics, disease characteristics and prior biologic therapy: an integrated analysis of the phase III UltIMMa-1 and UltIMMa-2 studies. J Eur Acad Dermatol Venereol. 2020;34(12):2830-2838. doi:10.1111/jdv.16521
- Strober B, Valdecantos WC, Zhan T, Lambert H, Menter A. Efficacy of risankizumab in moderate-to-severe plaque psoriasis by baseline characteristics and prior therapies. Presented at: Skin Inflammation & Psoriasis International Network (SPIN) 6th Congress; April 25–27, 2019; Paris, France. Poster P081.
- Data on File, AbbVie Inc. ABVRRTI71735.
- Keininger D, Coteur G. Assessment of self-injection experience in patients with rheumatoid arthritis: psychometric validation of the Self-Injection Assessment Questionnaire (SIAQ). Health Qual Life Outcomes. 2011;9:2. doi:10.1186/1477-7525-9-2
- ILUMETRI [Summary of Product Characteristics]. Almirall S.A.; May 2021.
- TREMFYA [Summary of Product Characteristics]. Janssen-Cilag International NV; January 2021.
- STELARA [Summary of Product Characteristics]. Janssen-Cilag International NV; May 2021.
- TALTZ [Summary of Product Characteristics]. Eli Lilly Nederland B.V.; August 2021.
- COSENTYX [Summary of Product Characteristics]. Novartis Europharm Limited; September 2021.
- BIMZELX [Summary of Product Characteristics]. UCB Pharma S.A.; August 2021.