Note to affiliates: This update to the venetoclax CLL AbbVie Pro site includes a homepage headline, updated CLL14 6-year, CLL13 4-year, MURANO 7-year data sets, and other streamlined content updates. CLL 13 4-year update reflects the CLL13 data from the Lancet Oncology publication. The CLL14 6-year and MURANO 7-year data have been updated based on the EHA 2023 abstracts. For countries that cannot use these data sets, please follow local regulations and MRLO guidance, and revert to CLL14 5-year and MURANO 5-year published data from the product label.
Primary analysis in ITT population for VEN+O vs O+Clb1:
INV-assessed PFS†: Reduced risk of progression or death (HR=0.35; 95% CI: 0.23–0.53 [P<0.0001]).
| • | Median follow-up of 28 months |
Additional analyses:
6-year PFS estimate (INV-assessed)2‡: 53% vs 22% (HR=0.40; 95% CI: 0.31–0.52) after 5 years off treatment.
| • | Median PFS of 76.2 months with VEN+O vs 36.4 months with O+Clb |
INV-assessed complete remission (CR/CRi)1: 50% vs 23% (P<0.0001).
| • | ORR: 85% (95% CI: 79.2–89.2) vs 71% (95% CI: 64.8–77.2 [P=0.0007]) |
Primary analysis in ITT population for VEN+R vs BR1:
INV-assessed PFS†: Reduced risk of progression or death (HR=0.17; 95% CI: 0.11–0.25 [P<0.0001]).
| • | Median follow-up of 23.8 months |
Additional analyses:
7-year PFS estimate (INV-assessed)3‡: 23% (HR=0.23; 95% CI: 0.18–0.29) vs NE after ~5 years off treatment.
| • | Median PFS of 54.7 months with VEN+R (95% CI: 52.3–59.9) vs 17.0 months with BR (95% CI: 15.5–21.7) |
INV-assessed complete remission (CR/CRi)1‡: 27% vs 8%.
| • | ORR: 93% (95% CI: 88.8–96.4) vs 68% (95% CI: 60.6–74.2) |
*See full dosing information for VEN+O and for VEN+R in the dosing and administration section.
†Primary endpoint.
‡Results are descriptive only.
1L=first line; CLL=chronic lymphocytic leukaemia; VEN+O=VENCLYXTO + obinutuzumab; ITT=intent to treat; O+Clb=obinutuzumab + chlorambucil; INV=investigator; PFS=progression-free survival; HR=hazard ratio; CI=confidence interval; CR=complete remission; CRi=complete remission with incomplete bone marrow recovery; ORR=overall response rate; 2L+=second line + later lines of therapy; VEN+R=VENCLYXTO + rituximab; BR=bendamustine + rituximab; NE=not evaluable.
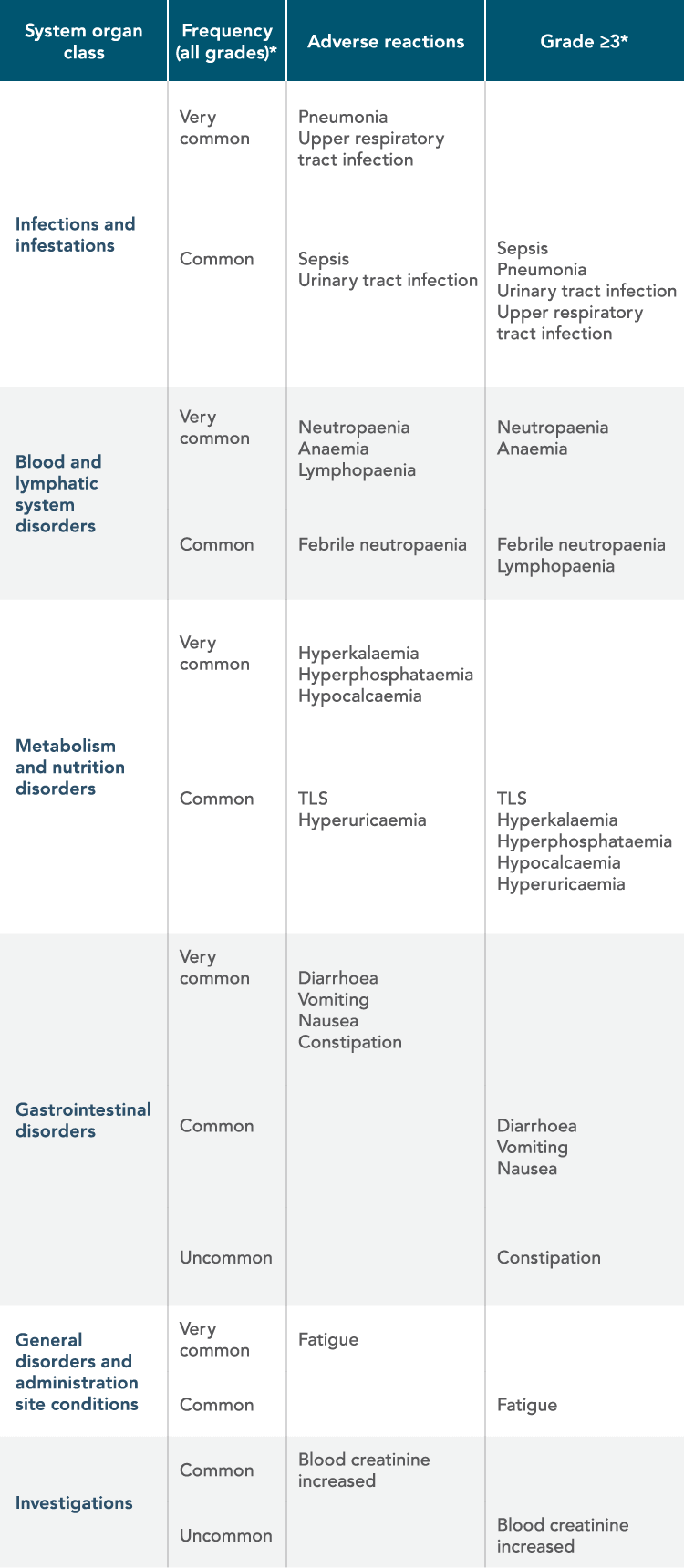
VENCLYXTO-BASED REGIMENS HAVE AN ESTABLISHED AND MANAGEABLE SAFETY PROFILE IN CLL CLINICAL TRIALS1
| • | The most frequently reported serious adverse reactions (≥2%) in patients receiving VENCLYXTO in combination with obinutuzumab or rituximab were pneumonia, sepsis, febrile neutropaenia, and TLS |
| • | The most commonly occurring adverse reactions (≥20%) of any grade in patients receiving VENCLYXTO in the combination studies with obinutuzumab or rituximab were neutropaenia, diarrhoea, and upper respiratory tract infection |
| ▼ | This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions. |
ADVERSE REACTIONS BY MedDRA BODY SYSTEM ORGAN CLASS AND BY FREQUENCY
*Only the highest frequency observed in the trials is reported (based on studies CLL14, MURANO, M13-982, M14-032, and M12-175).
TLS=tumour lysis syndrome; MedDRA=Medical Dictionary for Regulatory Activities.
Note to affiliate: the OPTIONAL content below is based on the “Al-Sawaf O, Zhang C, Tandon M, et al. Lancet Oncol 2020” publication and is not included in the SmPC. Use of this safety analysis should be evaluated by affiliate Med/Reg review based on local regulations and policy.
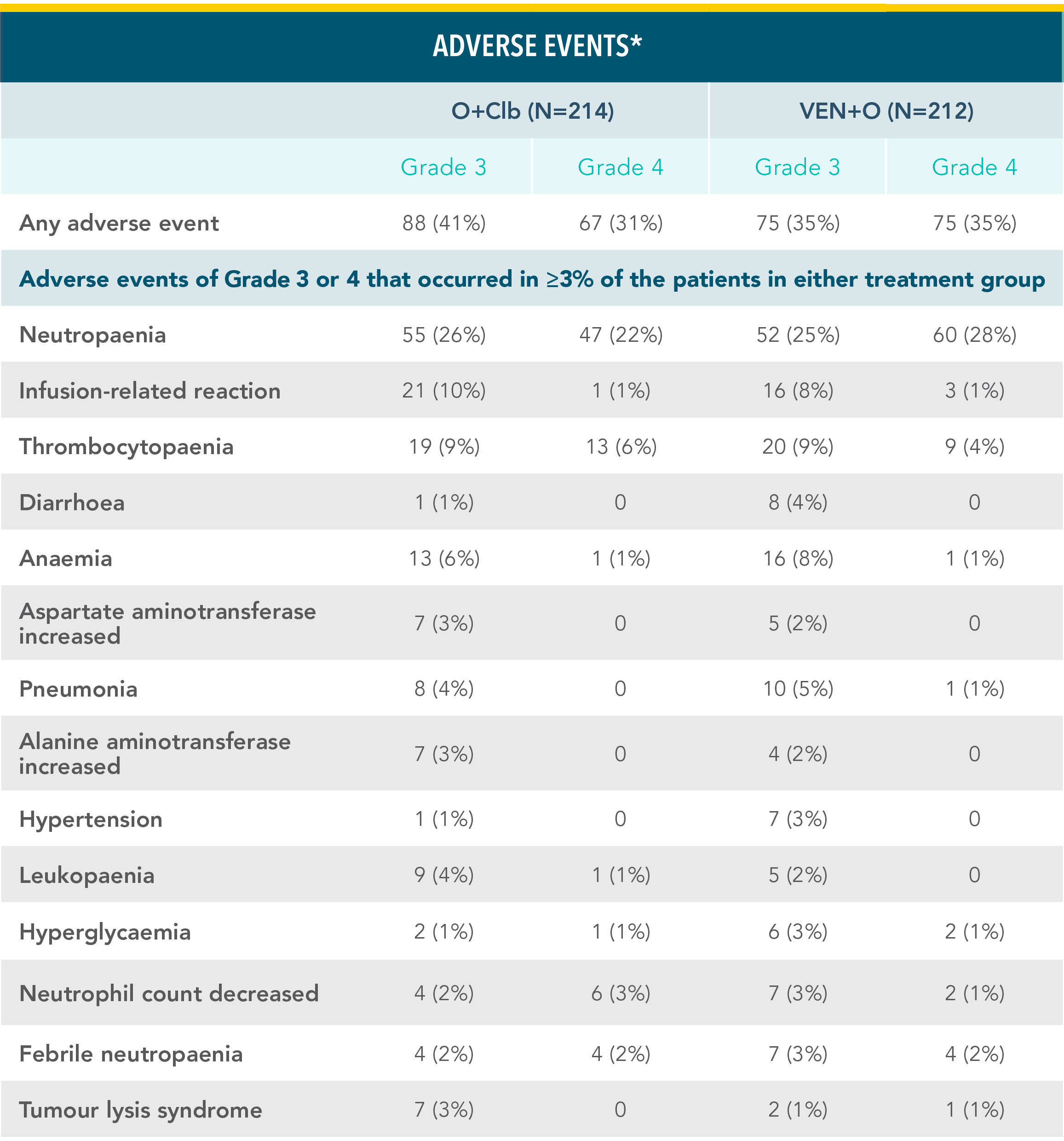
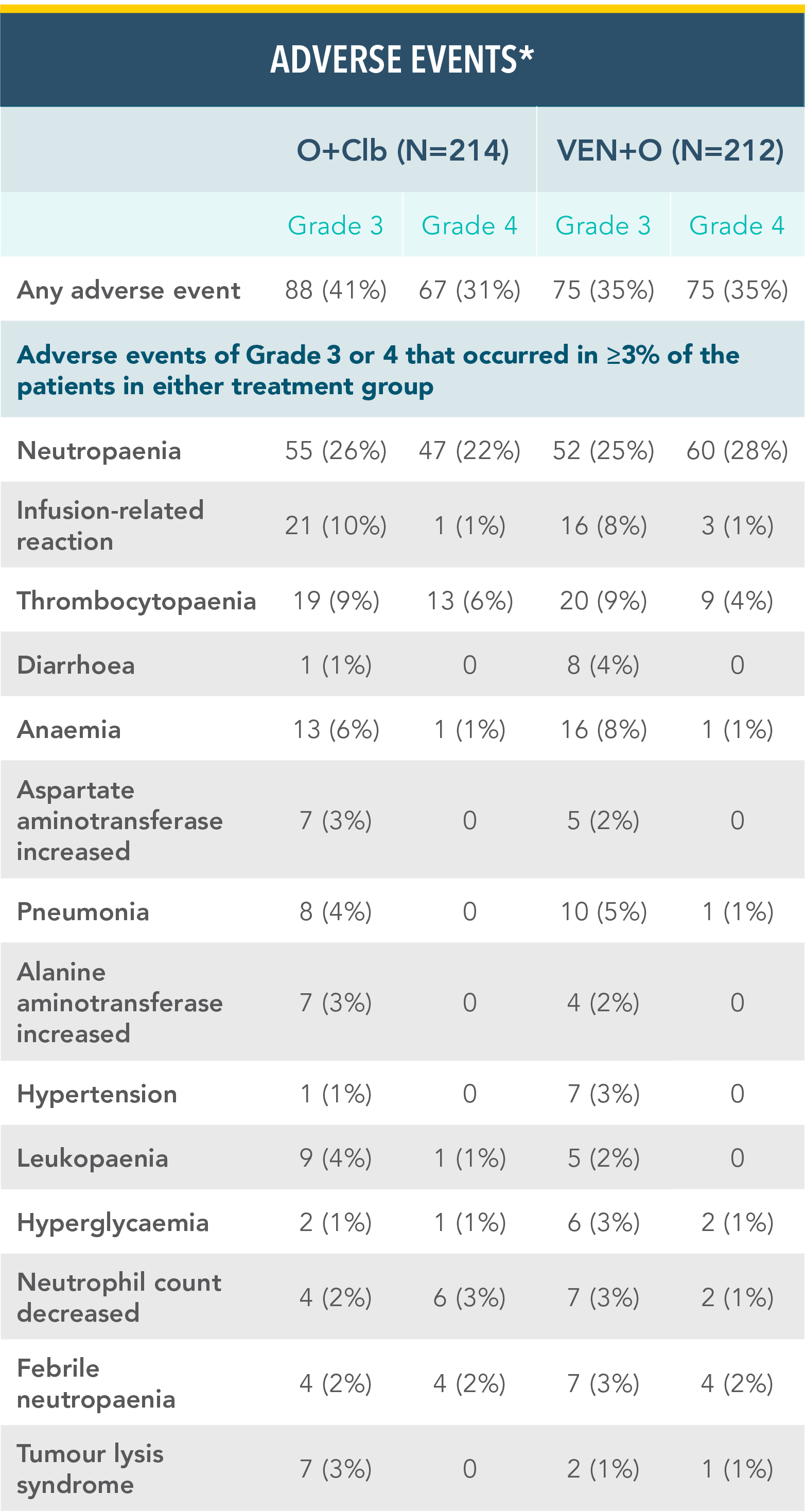
VENCLYXTO + OBINUTUZUMAB DEMONSTRATED A MANAGEABLE SAFETY PROFILE IN THE CLL14 TRIAL WITH 1 YEAR OF TREATMENTXX
Grade 3 or 4 adverse events in the VEN+O and O+Clb treatment armsxx
*Adverse events are reported according to Medical Dictionary for Regulatory Activities superclass and preferred terms and National Cancer Institute Common Terminology Criteria for Adverse Events 4.03 grade.
Reference: xxAl-Sawaf O, Zhang C, Tandon M, et al. Venetoclax plus obinutuzumab versus chlorambucil plus obinutuzumab for previously untreated chronic lymphocytic leukaemia (CLL14): follow-up results from a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2020;21(9):1188-1200.
Note to affiliate: the OPTIONAL content below includes cardiovascular adverse events from the CLL14 trial. It is based on the "Al-Sawaf O, Zhang C, Tandon M, et al. Lancet Oncol 2020” publication and is not included in the SmPC. Use of this page should be evaluated by affiliate Med/Reg review based on local regulations and policy.
ADDITIONAL SAFETY INFORMATION FROM THE CLL14 TRIALXX
Cardiovascular adverse events of Grade 1-2 with frequency of 10% or higher and/or Grade 3-5 with frequency of 1% or higher*
The analysis was not powered to demonstrate a statistically significant difference between VEN+O and O+Clb.
*Based on data as of clinical data cutoff date of August 23, 2019.
†Includes myocardial infarction and acute myocardial infarction.
Reference: xxAl-Sawaf O, Zhang C, Tandon M, et al. Venetoclax plus obinutuzumab versus chlorambucil plus obinutuzumab for previously untreated chronic lymphocytic leukaemia (CLL14): follow-up results from a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2020;21(9):1188-1200.
Note to affiliate: the OPTIONAL content below is based on the "Kater AP, Wu JQ, Kipps T, et al. J Clin Oncol 2020 supplementary appendix” publication and is not included in the SmPC. Use of this safety analysis should be evaluated by affiliate Med/Reg review based on local regulations and policy.
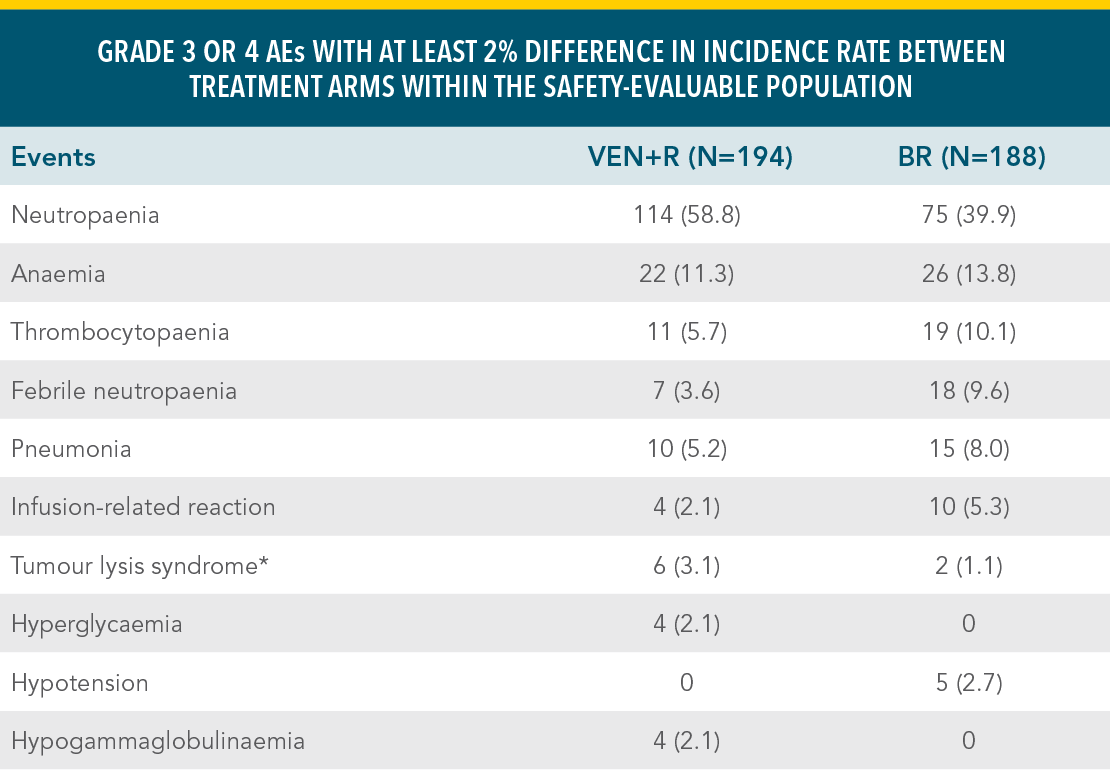
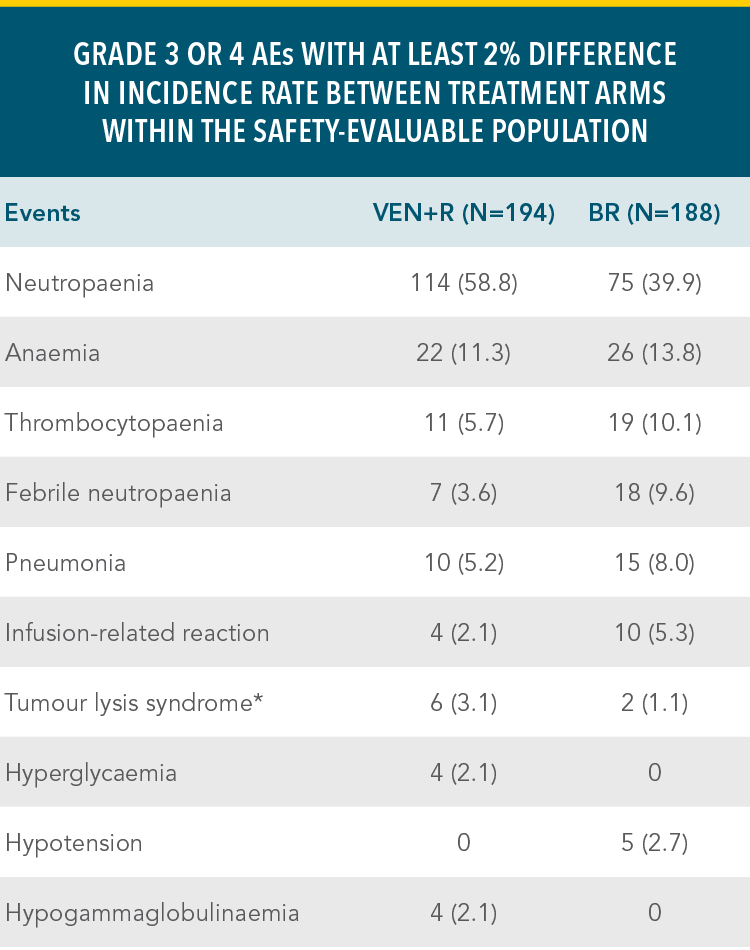
VENCLYXTO + RITUXIMAB DEMONSTRATED A MANAGEABLE SAFETY PROFILE IN THE MURANO TRIAL WITH 2 YEARS OF TREATMENTXX
Grade 3 or 4 adverse events in the VEN+R and BR treatment armsxx
| • | The duration of treatment was longer in the VEN+R arm than in the BR arm1 |
*Laboratory-confirmed TLS.
Reference: xxKater AP, Wu JQ, Kipps T, et al. Venetoclax plus rituximab in relapsed chronic lymphocytic leukemia: 4-year results and evaluation of impact of genomic complexity and gene mutations from the MURANO phase III study. J Clin Oncol. 2020;38(34):4042-4054(suppl appendix).
I want to find out
more
about VENCLYXTO
I want to receive more information about VENCLYXTO
Reference: 1. VENCLYXTO Summary of Product Characteristics. Ludwigshafen, Germany: AbbVie Deutschland GmbH & Co. KG.