INTEGRATED SAFETY ANALYSIS
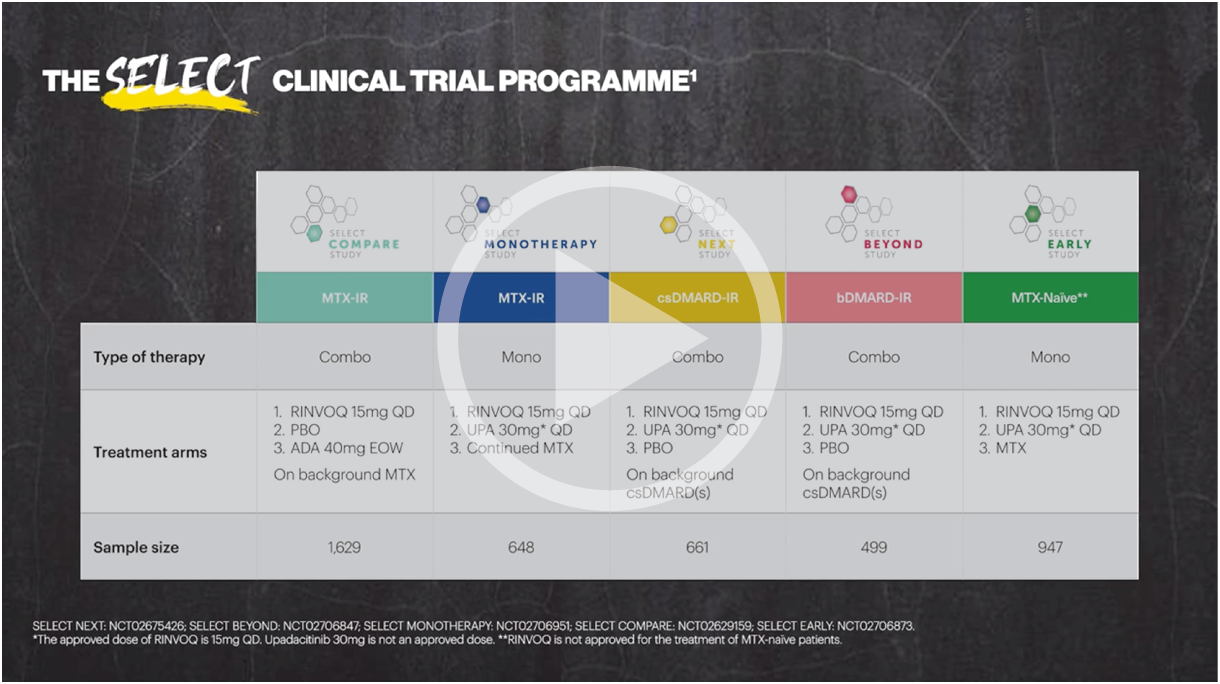
RINVOQ safety profile: Well-characterized in 5 Phase 3 studies
Integrated safety analysis across multiple patient populations up to 53 weeks of exposure1
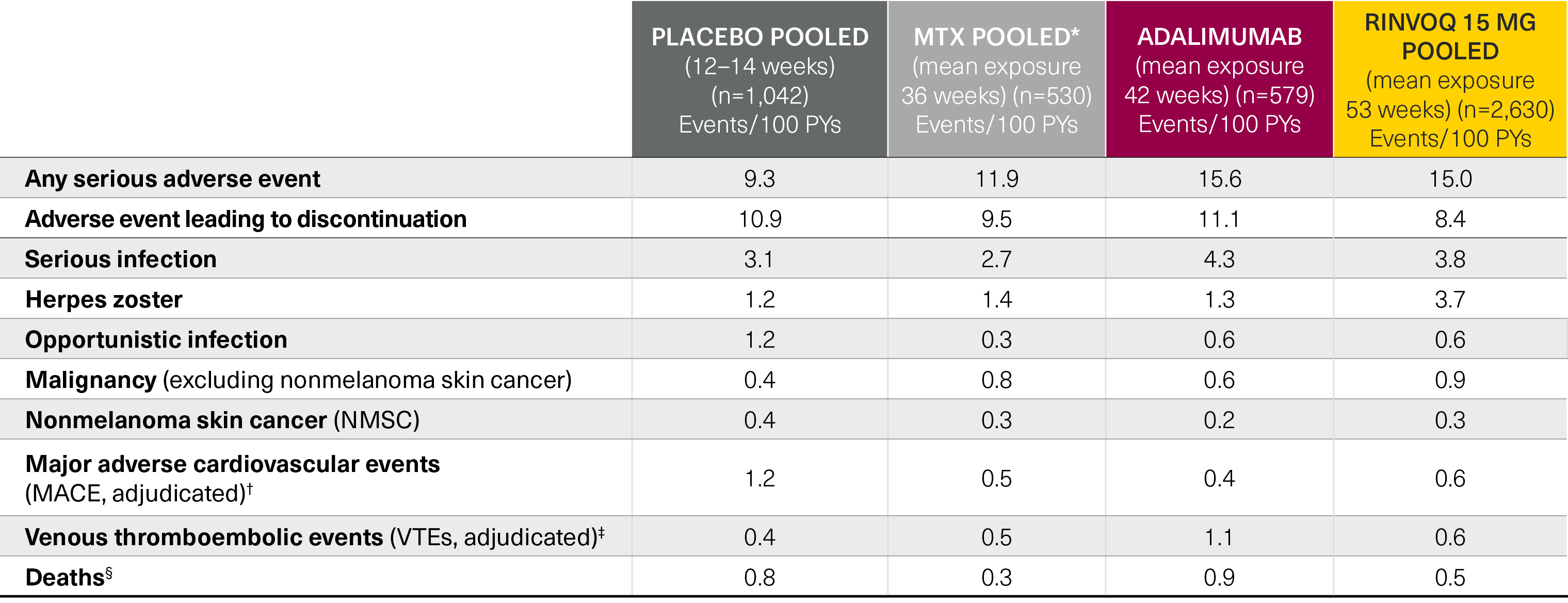
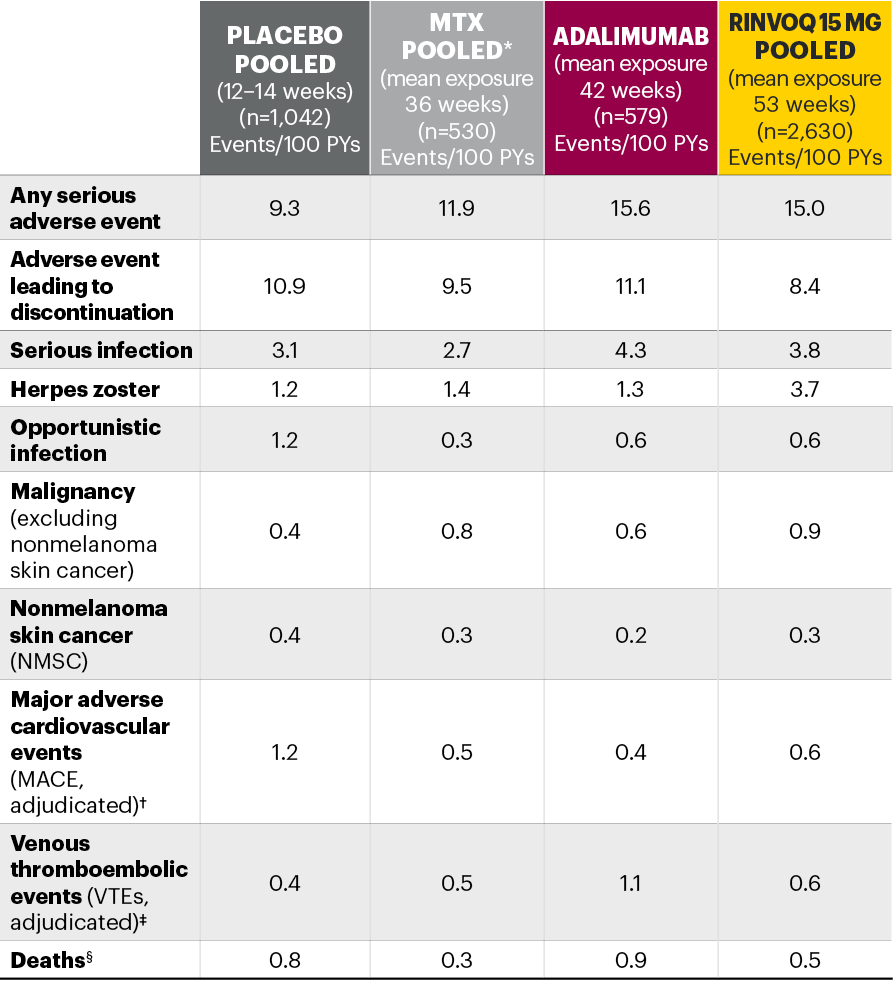
Five pivotal randomized, double-blind, controlled Phase 3 trials of RINVOQ were included in the integrated safety analyses. Placebo data were pooled from 3 placebo-controlled studies with exposures of up to 14 weeks (SELECT-NEXT and SELECT-BEYOND: up to 12 weeks; SELECT-COMPARE: up to 14 weeks). MTX data were pooled from 2 MTX-controlled studies (SELECT-EARLY and SELECT-MONOTHERAPY) with mean exposure of 36 weeks, including a maximum of 14 weeks of MTX treatment in SELECT-MONOTHERAPY. Originator adalimumab data were derived from 1 adalimumab-controlled study (SELECT-COMPARE) with mean exposure of 42 weeks. The RINVOQ 15 mg data were pooled from the above 5 studies with a mean exposure of 53 weeks. A total of 589 (22%) patients received RINVOQ for ≥72 weeks. Patients who switched from placebo, adalimumab, or MTX to RINVOQ were included in the RINVOQ analysis set from the start of RINVOQ, while those who switched from RINVOQ to adalimumab (SELECT-COMPARE) were included in the adalimumab data set from the start of adalimumab. All deaths and cardiovascular events across the clinical program were adjudicated by a blinded external cardiac adjudication committee. The most commonly reported adverse events in the RINVOQ 15 mg pooled group were upper respiratory tract infections, nasopharyngitis, and urinary tract infections.
*MTX pooled includes patients on MTX monotherapy censored at time of switch/addition of RINVOQ.
†MACE was defined as cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke.
‡VTE was defined as deep vein thrombosis and pulmonary embolism.
§Deaths included non–treatment-emergent deaths (3 on RINVOQ and 1 on adalimumab).
MTX: methotrexate; PYs: patient years.
Contraindications
- Hypersensitivity to the active substance or to any of the excipients
- Active tuberculosis (TB) or active serious infections
- Severe hepatic impairment
- Pregnancy
Summary of the RINVOQ safety profile
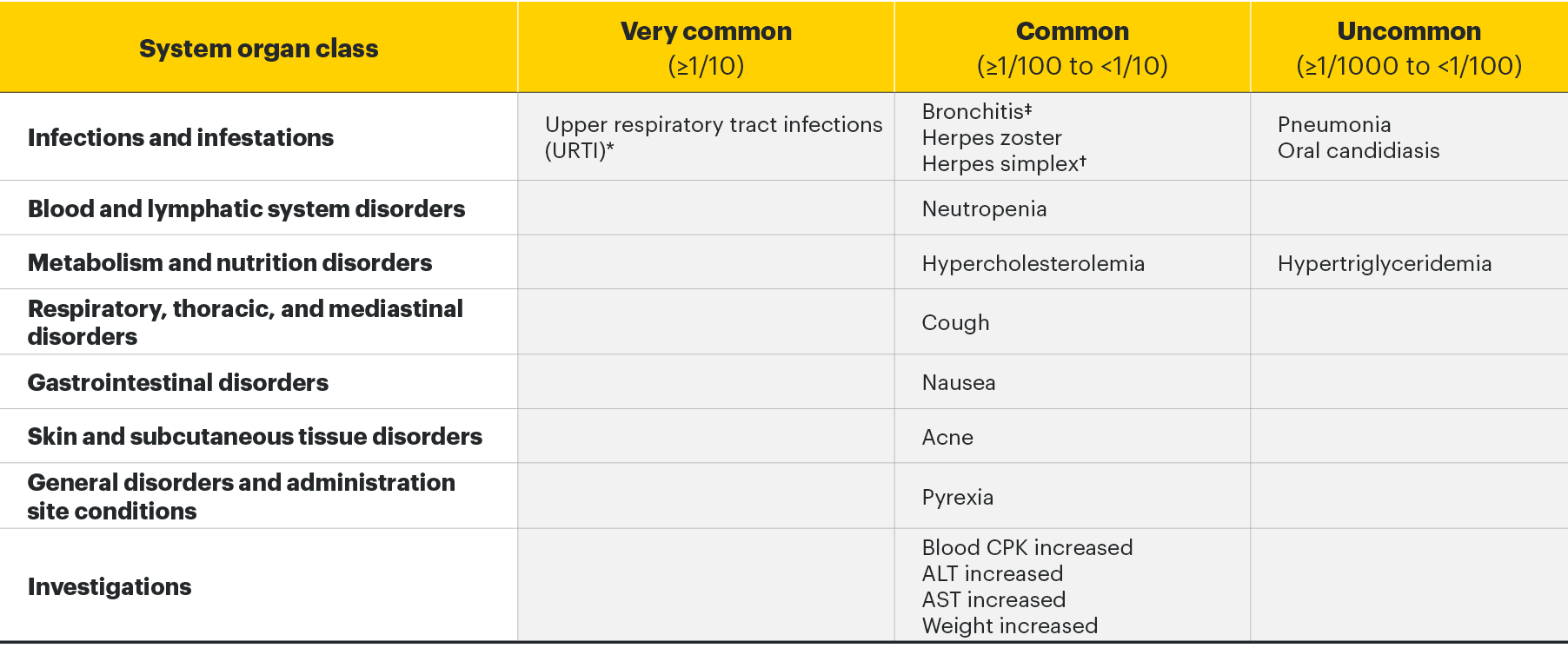
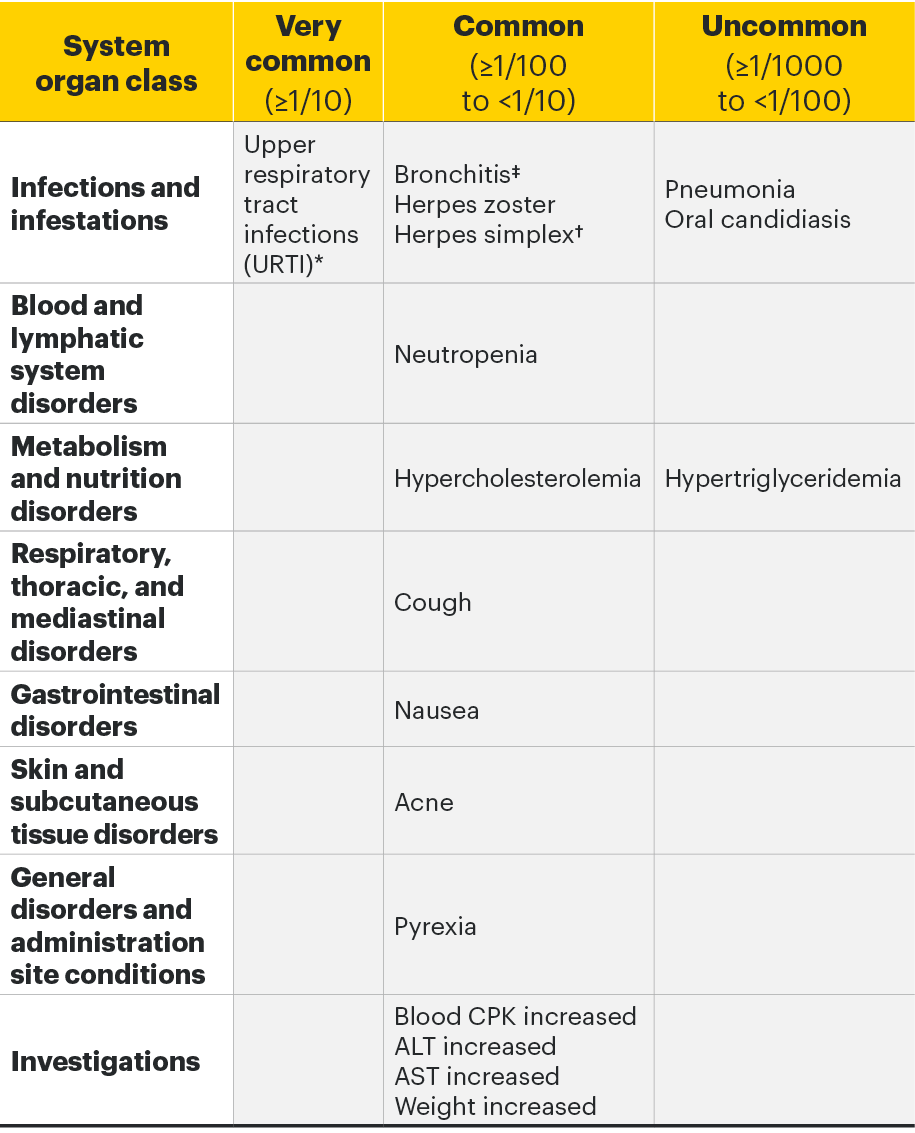
The most commonly reported adverse drug reactions (ADRs) were upper respiratory tract infections, bronchitis, nausea, blood creatine phosphokinase (CPK) increased, and cough. The most common serious adverse reactions were serious infections.2
Adverse drug reactions2
The following table of adverse reactions is based on experience from registrational clinical studies.
Within each frequency grouping, undesirable effects are presented in order of decreasing seriousness.
*Includes upper respiratory tract infection, acute sinusitis, laryngitis, nasopharyngitis, oropharyngeal pain, pharyngitis, pharyngotonsillitis, rhinitis, sinusitis, tonsillitis, viral upper respiratory tract infection.
†Includes herpes simplex and oral herpes.
‡Includes bronchitis, bronchitis viral, bronchitis bacterial, and tracheobronchitis.
For more detailed safety information, please see the Important Safety Information link at the top of the page, or for complete prescribing information, the RINVOQ SmPC.
Special Warnings and Precautions2
- Serious infections: RINVOQ should not be initiated in patients with an active, serious infection, including localized infections. Closely monitor patients for the development of signs and symptoms of infection during and after treatment with RINVOQ. If a patient develops a serious or opportunistic infection, RINVOQ should be interrupted. As there is a higher incidence of infections in the elderly ≥65 years of age, caution should be used when treating this population.
- Tuberculosis: Patients should be screened for tuberculosis (TB) before starting RINVOQ. Treatment should not be initiated in patients with active TB. Anti-TB therapy should be considered prior to initiation of RINVOQ in patients with previously untreated latent TB or in patients with risk factors for TB infection. Monitor patients for the development of signs and symptoms of TB, including patients who tested negative for latent TB infection prior to initiating therapy.
- Viral reactivation: Screening for viral hepatitis and monitoring for reactivation should be performed before starting and during treatment with RINVOQ. The risk of herpes zoster appears to be higher in Japanese patients treated with upadacitinib. If a patient develops herpes zoster, consider interruption of RINVOQ therapy until the episode resolves. If hepatitis B virus DNA is detected while receiving RINVOQ, a liver specialist should be consulted.
- Nonmelanoma skin cancer: Periodic skin examination is recommended for patients who are at increased risk for skin cancer.
- Vaccinations: Use of live, attenuated vaccines during or immediately prior to RINVOQ treatment is not recommended. Prior to initiating RINVOQ, it is recommended that patients be brought up to date with all immunizations, including prophylactic zoster vaccinations, in agreement with current immunization guidelines.
- Venous thromboembolism: RINVOQ should be used with caution in patients at high risk for deep venous thrombosis (DVT)/pulmonary embolism (PE). If clinical features of DVT/PE occur, RINVOQ treatment should be discontinued and patients should be evaluated promptly, followed by appropriate treatment.
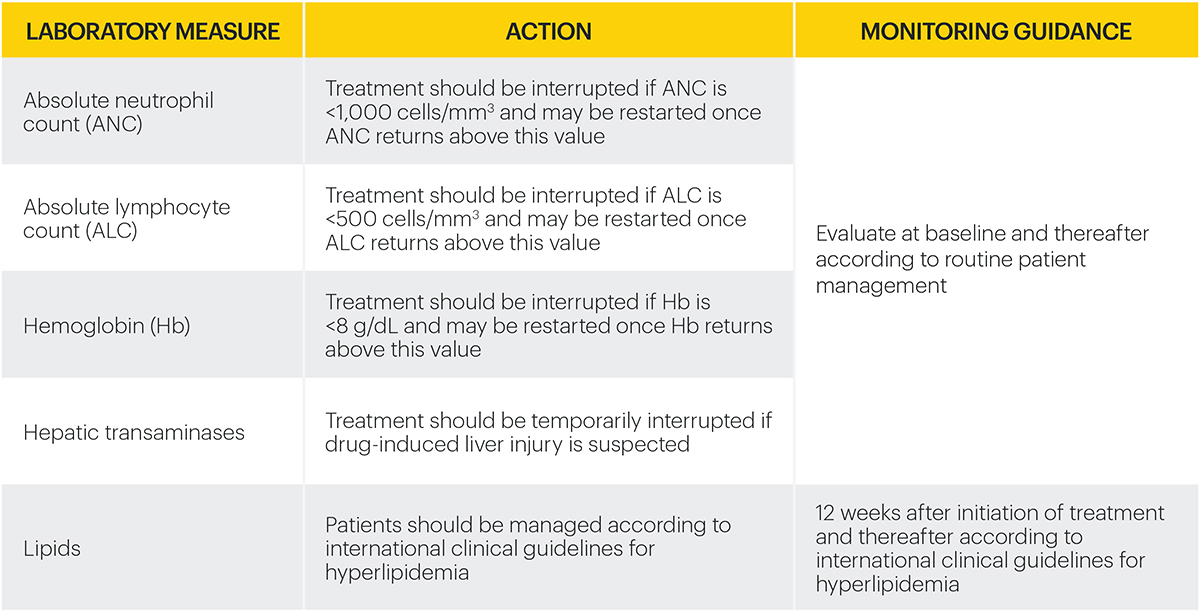
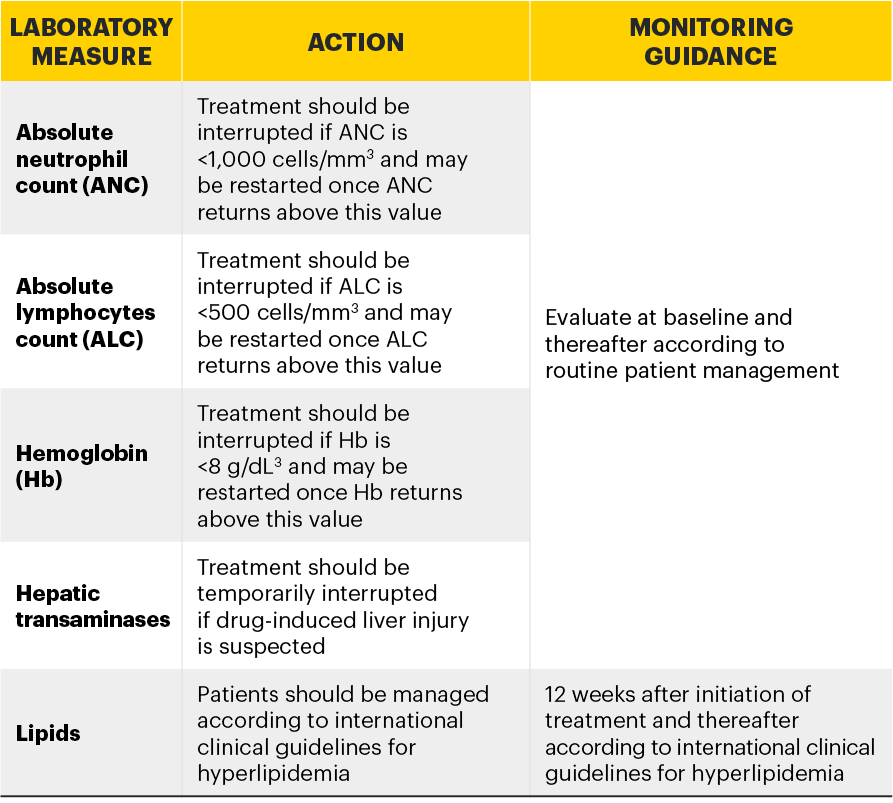
Monitoring requirements
Treatment with RINVOQ should not be initiated if:2
- Absolute neutrophil count <1,000 cells/mm3*
- Absolute lymphocyte count <500 cells/mm3*
- Hemoglobin <8 g/dL*
Laboratory measures and monitoring guidance2
Elderly
No dose adjustment is required in patients aged 65 years and older. There are limited data in patients aged 75 years and older.
Renal impairment
No dose adjustment is required in patients with mild or moderate renal impairment. There are limited data on the use of RINVOQ in subjects with severe renal impairment. RINVOQ should be used with caution in patients with severe renal impairment. The use of upadacitinib has not been studied in subjects with end-stage renal disease.2
Hepatic impairment
No dose adjustment is required in patients with mild (Child Pugh A) or moderate (Child Pugh B) hepatic impairment. RINVOQ should not be used in patients with severe (Child Pugh C) hepatic impairment.2
Pediatric
The safety and efficacy of RINVOQ in children and adolescents aged 0 to less than 18 years have not yet been established. No data are available.
*Treatment may be initiated or restarted after levels return above specified values.
Safety information2
References
- Cohen SB, van Vollenhoven RF, Winthrop KL, et al. Safety profile of upadacitinib in rheumatoid arthritis: integrated analysis from the SELECT phase III clinical programme. Ann Rheum Dis. 2020;80(3):304-311. doi:10.1136/annrheumdis-2020-218510
- RINVOQ [Summary of Product Characteristics]. AbbVie Deutschland GmbH & Co. KG; May 2021.