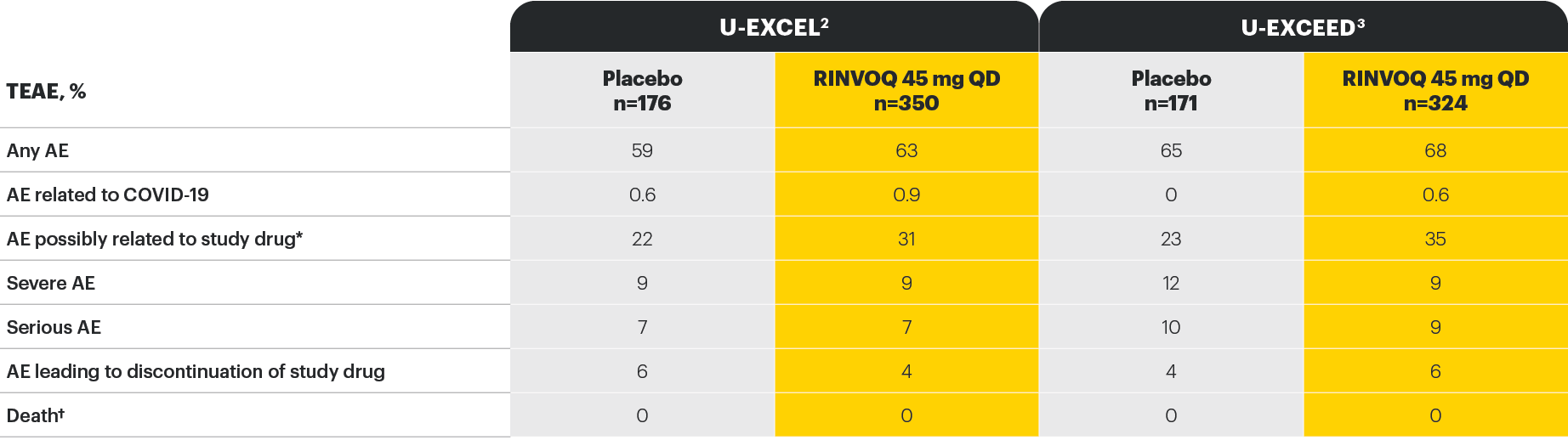
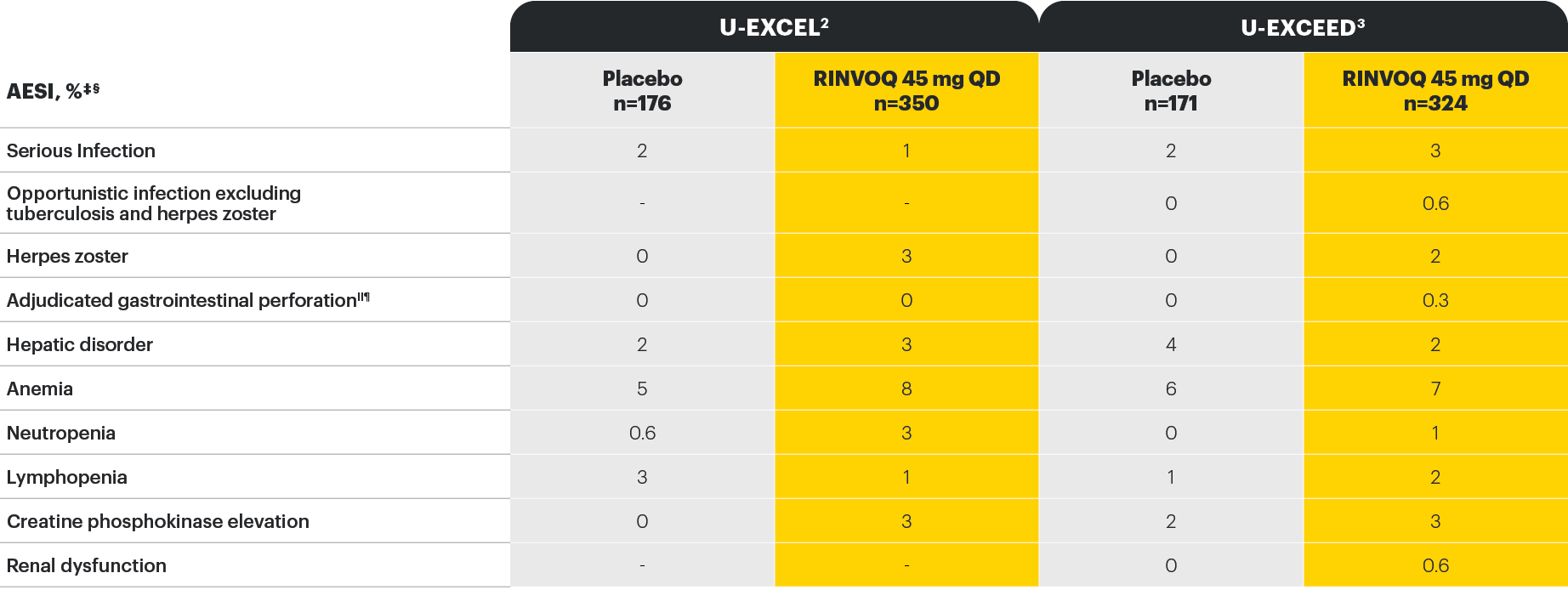
Well-studied safety profile in Crohn’s disease through Induction Week 12
*As assessed by investigator.
†In U-EXCEED, there was one non-treatment emergent death due to infectious shock in a patient who received RINVOQ 45 mg QD for 5 days; the death occurred 159 days after premature discontinuation from the study.
‡In U-EXCEL, no opportunistic infections (excluding tuberculosis and herpes zoster), tuberculosis, renal disorders, adjudicated cardiovascular or venous thromboembolic events, or cancer of any kind was observed in either group.
§In U-EXCEED, no tuberculosis, major adverse cardiovascular events, adjudicated venous thromboembolic events, or cancer of any kind was observed in either group.
||In U-EXCEL, one event of adjudicated gastrointestinal perforation (intestinal perforation) was reported in a patient who was a clinical nonresponder to placebo and was on RINVOQ 45 mg QD in the extended treatment period.
¶In U-EXCEED, 2 additional events of adjudicated gastrointestinal perforation (retroperitoneal abscess and ileal perforation) were reported in patients who were nonresponders to placebo and were on RINVOQ 45 mg QD in the extended treatment period.
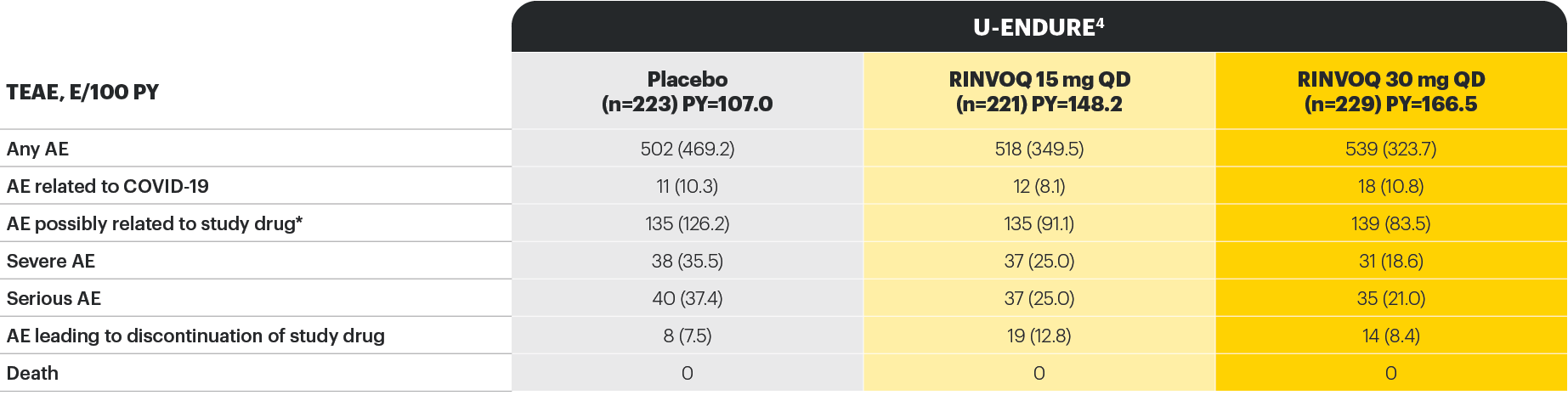
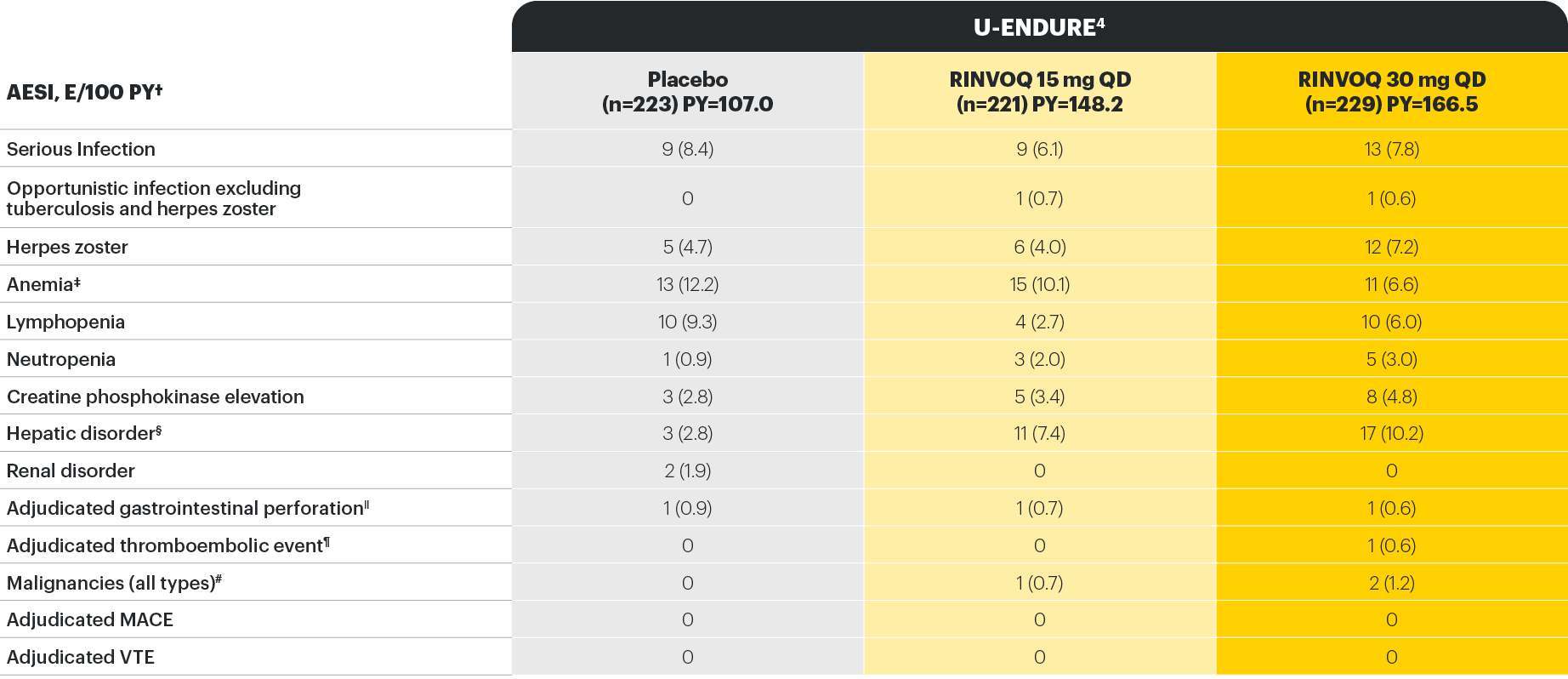
Well-studied safety profile in Crohn’s disease through Maintenance Week 52
*As assessed by investigator.
†No tuberculosis, adjudicated cardiovascular, or nonmelanoma skin cancer events were observed in any treatment group.
‡Anemia (which includes other preferred terms, in addition to the preferred term “anaemia”), herpes zoster, neutropenia, and lymphopenia were based on CMQ search.
§Hepatic disorder included transaminase elevations that were mild or moderate, asymptomatic, nonserious, and uncommonly led to treatment discontinuation.
||In all patients treated with rescue RINVOQ 30 mg QD (n=336), gastrointestinal perforation was reported in 3 patients (0.8 per 100 patient-years).
¶Hepatic vein thrombosis concurrent with an event of exacerbation of CD.
#Metastatic ovarian cancer in a patient in the RINVOQ 15 mg group and colon cancer and invasive lobular breast cancer in one patient each in the RINVOQ 30 mg QD group.
AE: adverse event; AESI: adverse event of special interest; APS: abdominal pain score; CDAI: Crohn’s Disease Activity Index; JAK: Janus kinase; MACE: major adverse cardiac event; PY: patient-year; QD: once daily; SES-CD: simple endoscopic activity score for Crohn’s disease; SF: stool frequency; TEAE: treatment-emergent adverse event; VTE: venous thromboembolism.
Study designs: the U-EXCEL and U-EXCEED induction studies were both multicenter, double-blind, placebo-controlled clinical studies. In U-EXCEL (N=526 [287 bio-naive, 239 biologic failures]) and U-EXCEED (N=495 biologic failures only), patients were randomized to RINVOQ 45 mg QD or placebo for 12 weeks with a 2:1 treatment allocation ratio and included in the efficacy analysis. In both studies, induction nonresponders were allowed to enter an additional 12-week open-label extended treatment period. All enrolled patients had moderately to severely active CD defined as SF ≥4 and/or APS ≥2, plus an SES-CD ≥6 (≥4 for patients with isolated ileal disease) excluding the narrowing component. U-ENDURE maintenance was a multicenter, double-blind, placebo-controlled clinical study with 502 patients who achieved clinical response (≥30% decrease in average daily SF and/or in APS, neither worse than baseline) to 12 weeks of RINVOQ 45 mg QD induction treatment. These patients were rerandomized 1:1:1 to receive either RINVOQ 15 mg QD, 30 mg QD, or placebo.1
UP NEXT
[Affiliates to insert local summary of safety]
REFERENCES
- RINVOQ [Summary of Product Characteristics]. AbbVie Deutschland GmbH & Co. KG; April 2023.
- Loftus EV Jr, Colombel JF, Lacerda AP, et al. Efficacy and safety of upadacitinib induction therapy in patients with moderately to severely active Crohn’s disease: results from a randomized phase 3 U-EXCEL study. Presented at: United European Gastroenterology Week; October 8-11, 2022; Vienna, Austria.
- Colombel JF, Panes J, Lacerda AP, et al. Efficacy and safety of upadacitinib induction therapy in patients with moderately to severely active Crohn’s disease who failed prior biologics: results from the randomized phase 3 U-EXCEED study. Presented at: Digestive Disease Week; May 21-24, 2022; San Diego, CA.
- Panes J, Loftus EV Jr, Lacerda AP, et al. Efficacy and safety of upadacitinib maintenance therapy in patients with moderately to severely active Crohn’s disease: results from a randomized phase 3 U-ENDURE maintenance study. Presented at: American College of Gastroenterology Annual Meeting; October 21-26, 2022; Charlotte, NC.