[Note to Affiliates: HCP-delivered patient materials and their inclusion herein are subject to local codes and regulations.]
[ NEW VIDEOS TO COME ]
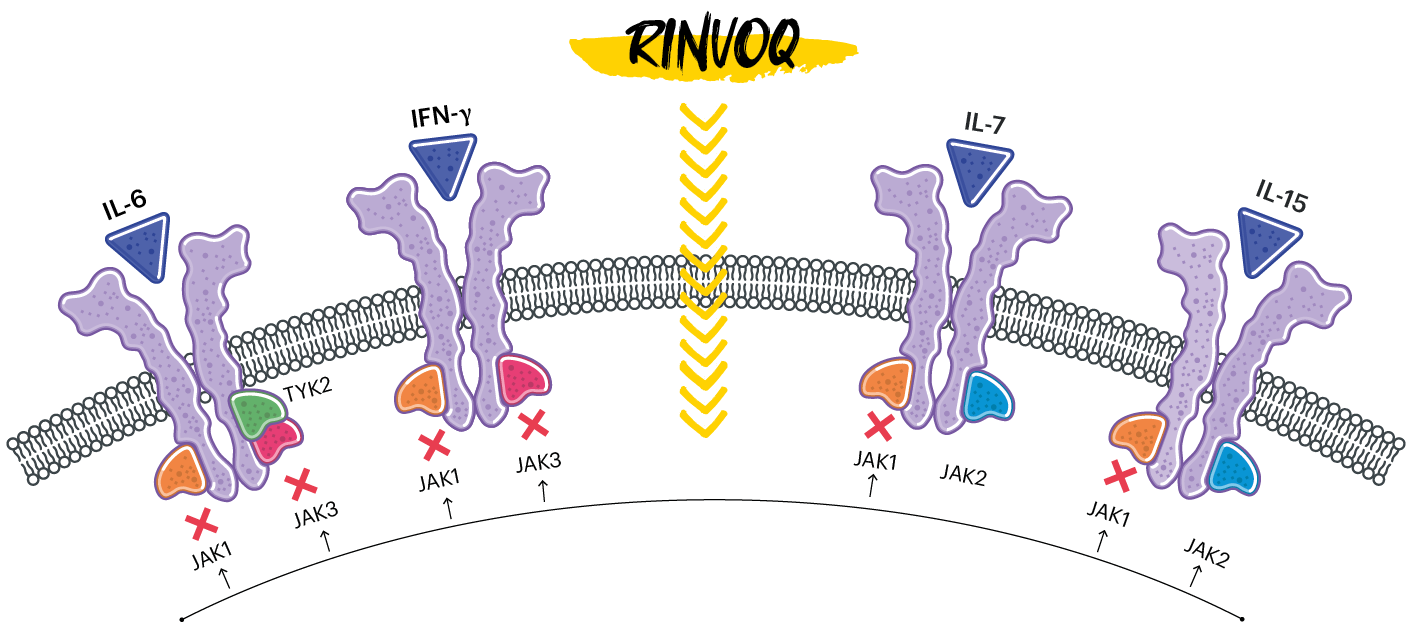
RINVOQ IS A SELECTIVE AND REVERSIBLE JAK INHIBITOR1
In human cellular assays, RINVOQ preferentially inhibits signaling by JAK1 or JAK1/3 with functional selectivity over cytokine receptors that signal via pairs of JAK2.1
(RINVOQ's JAK inhibition is represented by X)
*In vitro potency in engineered cellular assays. Fold selectivity is indicative of how much more selective RINVOQ is to JAK1 over other JAKs.
[Affiliate to create and gain local MLOR approval of this PDF, including ISI]
APS: abdominal pain score; JAK: Janus kinase; IL: interleukin; SES-CD: simple endoscopic activity score for Crohn’s disease; SF: stool frequency.
Study designs: the U-EXCEL and U-EXCEED induction studies were both multicenter, double-blind, placebo-controlled clinical studies. In U-EXCEL (N=526 [287 bio-naive, 239 biologic failures]) and U-EXCEED (N=495 biologic failures only), patients were randomized to RINVOQ 45 mg Q or placebo for 12 weeks with a 2:1 treatment allocation ratio and included in the efficacy analysis. In both studies, induction nonresponders were allowed to enter an additional 12-week open-label extended treatment period. All enrolled patients had moderately to severely active CD defined as SF ≥4 and/or APS ≥2, plus an SES-CD ≥6 (≥4 for patients with isolated ileal disease) excluding the narrowing component. U-ENDURE maintenance was a multicenter, double-blind, placebo-controlled clinical study with 502 patients who achieved clinical response (≥30% decrease in average daily SF and/or in APS, neither worse than baseline) to 12 weeks of RINVOQ 45 mg QD induction treatment. These patients were rerandomized 1:1:1 to receive either RINVOQ 15 mg QD, 30 mg QD, or placebo.1
LOOKING FOR MORE INFORMATION?
[Affiliates to insert local summary of safety]
REFERENCES
- RINVOQ [Summary of Product Characteristics]. AbbVie Deutschland GmbH & Co. KG; April 2023.
- Parmentier JM, Voss J, Graff C, et al. In vitro and in vivo characterization of the JAK1 selectivity of upadacitinib (ABT-494). BMC Rheumatol. 2018;2:23. doi:10.1186/s41927-018-0031-x