Find out more about the efficacy of SKYRIZI
Explore the durability of response with SKYRIZI through clinical trials and long-term data in psoriasis and psoriatic arthritis. Start by selecting a study from the menu below.
LONG-TERM DATA
HEAD-TO-HEAD TRIALS
SKYRIZI IN PsA
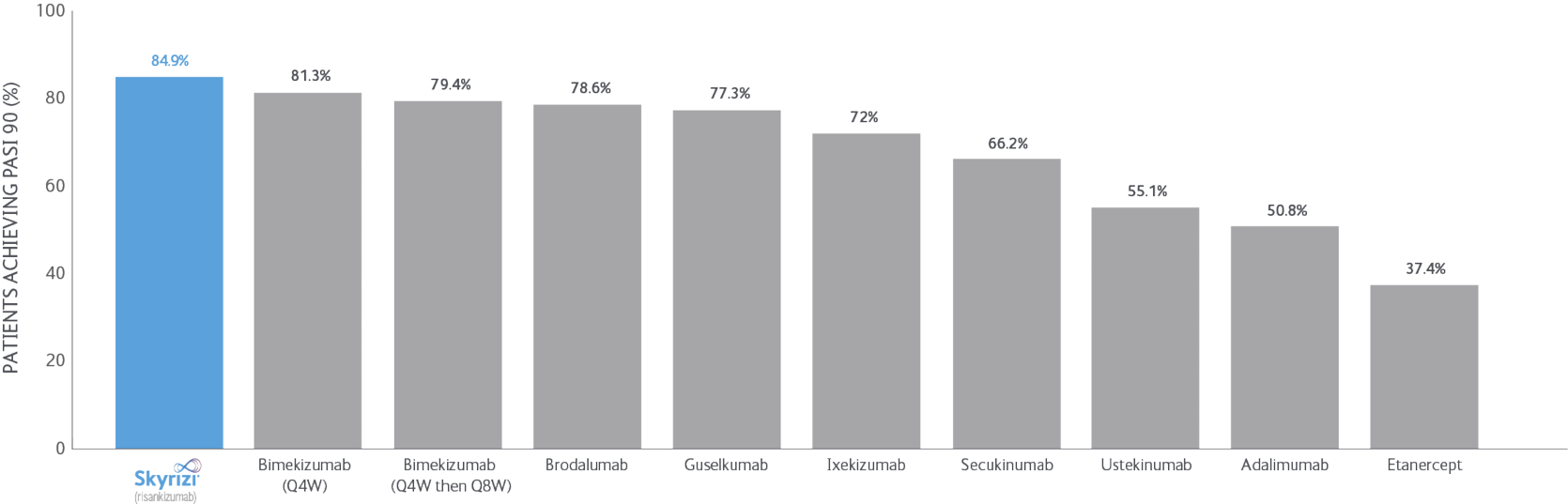
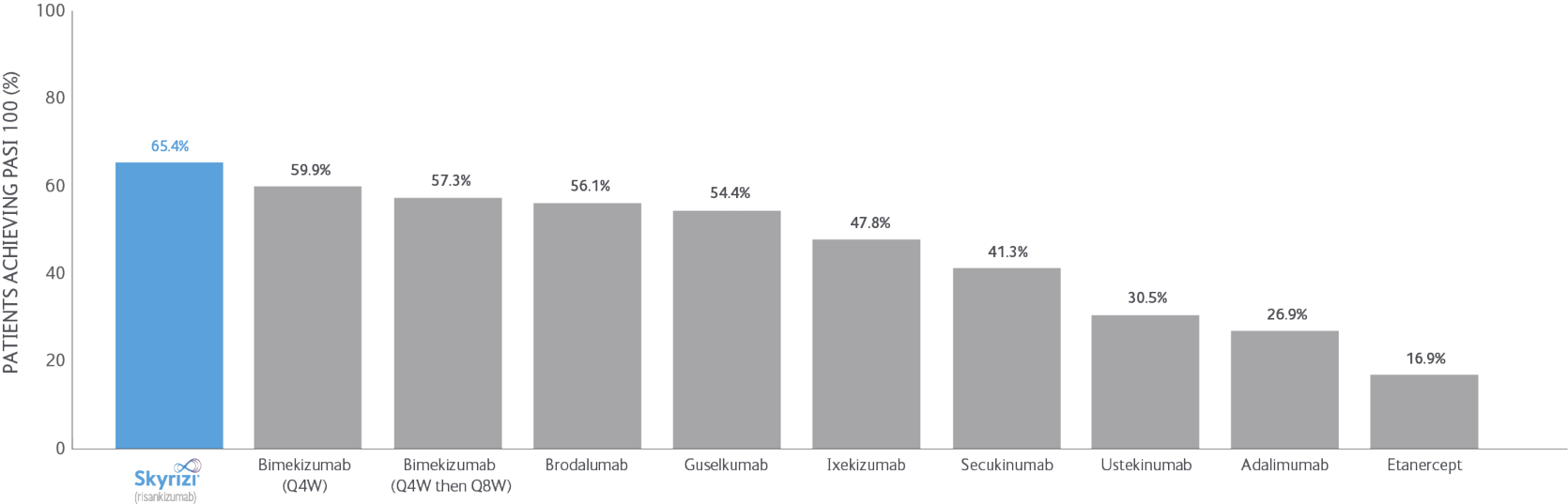
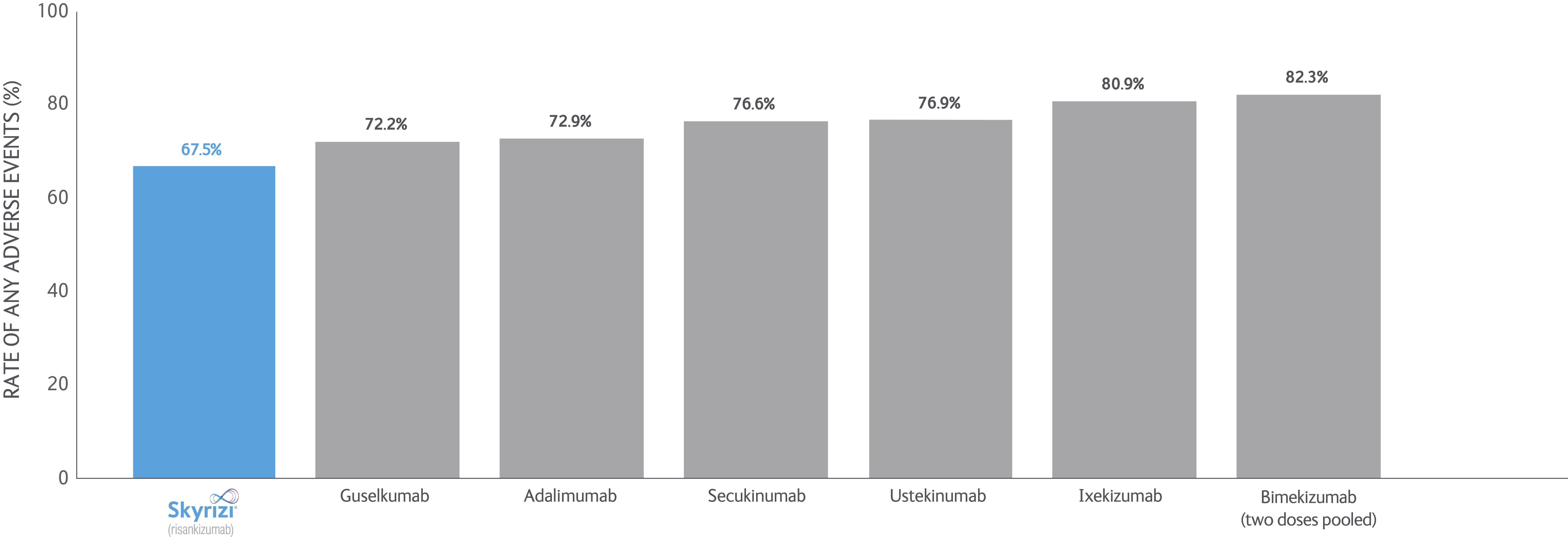
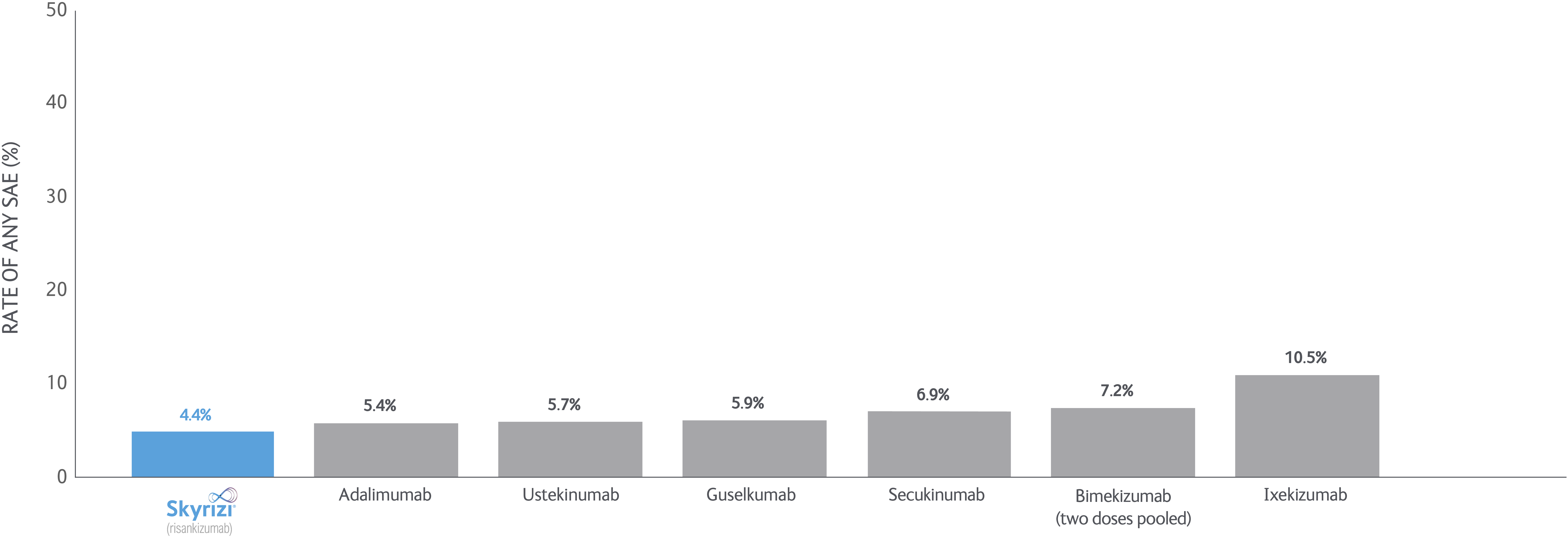
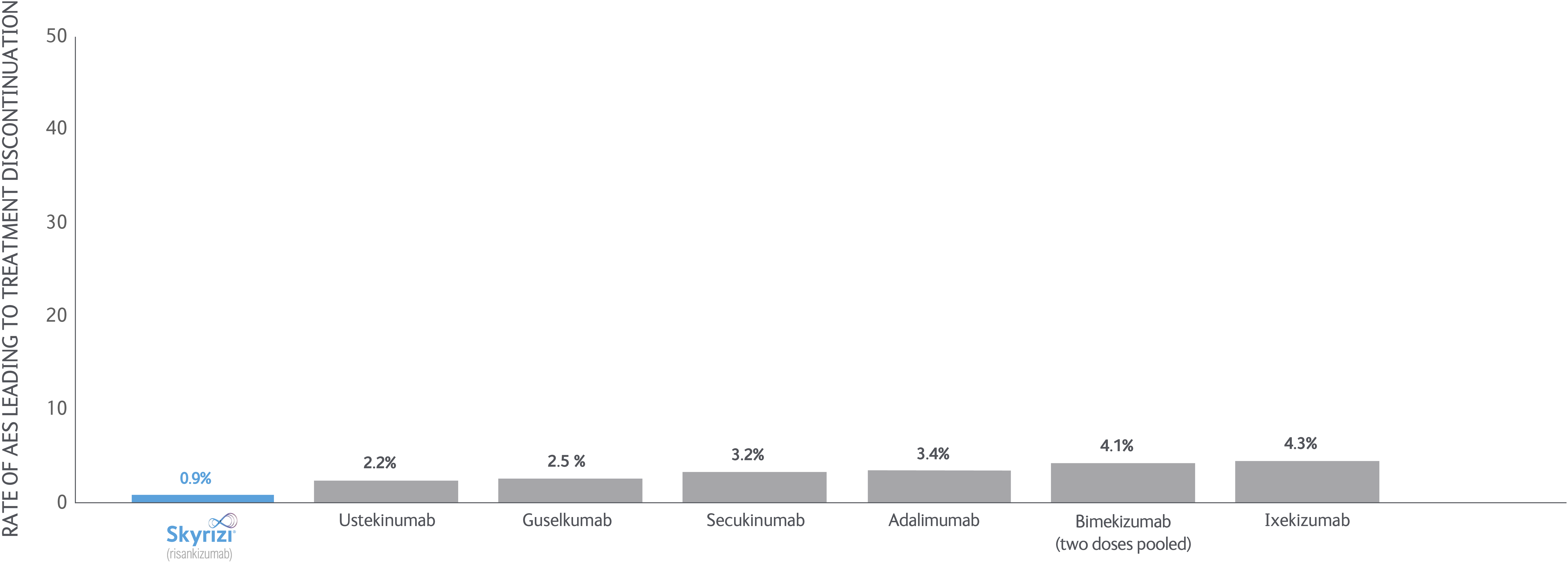
SKYRIZI LONG-TERM DATA: NMA data
Limitations of NMAs
Network meta-analyses simultaneously analyse all direct and indirect evidence for comparisons of treatments across the network. The main consideration for limitations in a network meta-analysis are to ensure that the relative contributions of different sources of direct evidence, which may have different study limitations, are accounted for appropriately. Judgments should be made across comparisons, considering potential differences between the populations, treatments, and outcomes by the network meta-analysis.
Limitations of this study
The current analysis is limited to outcomes at 48-56 weeks. This analysis is based on data obtained from 215 RCTs from 689 publications. The results need to be validated using real-world data that reflect actual utilisation and outcomes patterns in the psoriasis population to evaluate its generalisability.
Objectives
Bayesian network meta-analyses (NMs) were conducted to compare the efficacy (at least a 75/90/100% reduction in PASI score from baseline) and safety outcomes (any adverse event (AE), any serious AE (SAE), and AEs loading to treatment discontinuation) of each treatment evaluated between Weeks 48 and 56 after baseline.
This systematic literature review was originally conducted on December 4,2017, and updated on September 17, 2018, February 19, 2021, and May 2, 2021. The 2021 update incorporates recently licensed biologics in Europe and the US, covering all doses published in randomised controlled trials (RCTs).
Study selection
215 RCTs from 689 publications through May 2, 2021. A total of 14 studies were included in the MAs of long-term efficacy and safety outcomes, while 201 were excluded. The searched databases include Embase, MEDLINE, and the Cochrane library. Additional searches were conducted for the reference lists of included studies, conference proceedings, previous health technology assessment submissions, and clinical trial registries.
Data analysis
Rankings were quantified by the surface under the cumulative ranking curve (SUCRA). The benefit-risk profiles of treatments were assessed by bidimensional plots of the NMA-estimated efficacy and safety outcomes.
Study funding
AbbVie Inc funded the study and participated in the study design, research, analysis, data collection, interpretation of data, review, and approval of the publication. No honoraria or payments were made for authorship.
Select definitions
SUCRA: The surface under the cumulative ranking curve, a simple transformation of the mean rank, is used to provide a hierarchy of the treatments and accounts both for the location and the variance of all relative treatment effects. The larger the SUCRA value, the better the rank of the treatment.
Clear/nearly clear: At least a 75/90/100% reduction in PSI score from baseline improvement in PASI.
Tolerability: Withdrawal due to any adverse event.
Featured content
The Psoriasis Patient with Comorbidities Revisited
Journey back and see how a 53-year-old male with multiple comorbidities and chronic plaque psoriasis since 1981 achieved skin clearance with SKYRIZI for over 3 years, allowing him to pursue his interests without the disruption of frequent clinic visits.
UK-RISN-260022. Date of preparation April 2026.
References
- Reich K, et al. Lancet 2019; 394: 576-586.
- Gordon KB, et al. Lancet 2018; 392: 650-661.
- Warren RB, et al. Br J Dermatol 2021;184:50-59
- SKYRIZI: Summary of Product Characteristics.
- Armstrong AW, et al. Dermatol Ther 2022; 12:167-184.
UK-RISN-260042. Date of preparation: April 2026.