Timing matters: Bring device-aided therapies (DATs) early into the conversation1,2
How DATs can step in
Most people with PD are managed with levodopa and other oral or non-oral transdermal drug therapies. However, in advanced patients with difficult-to-manage motor fluctuations and dyskinesias, device-assisted therapies (DATs) may be beneficial. DATs for PD include apomorphine continuous subcutaneous infusion, levodopa continuous intestinal gel infusion, levodopa continuous subcutaneous infusion and deep brain stimulation.3
Use of DATs was examined in the PROSPECT study, a 24-month international, prospective, observational study of adults (≥30 years) with idiopathic PD and inadequately controlled motor fluctuations.* The study evaluated the long-term evolution of clinical outcomes and treatment patterns in individuals with advanced PD whose symptoms were insufficiently controlled with their existing therapy. The primary endpoint was the ‘OFF’ time change from baseline to Month 24, assessed by PD diaries.4,5
Real world evidence is collected outside of controlled clinical trials and has inherent limitations including a lesser ability to control for confounding factors.
PROSPECT4,5
Study type: Prospective observational study.
Aim: PROSPECT (PRospective Observational Study to evaluate the disease Progression and burdEn of disease of PD patients inadequately Controlled by conventional Therapy) aimed to evaluate long-term clinical outcomes and disease burden in people with advanced PD with motor fluctuations whose symptoms are inadequately controlled by their current medications and who have not yet initiated a device-aided therapy. The primary endpoint was the ‘OFF’ time change from baseline to month 24, assessed by PD diaries. Secondary endpoints included the change from baseline in Hoehn & Yahr (HY) stage for disease severity, Movement Disorder Society-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) part II for activities of daily living, 39-Item Parkinson’s disease questionnaire (PDQ-39) for quality of life, and non-motor symptom scale (NMSS). At enrolment, patients with advanced PD could not be treated with any device-aided therapy. Outcomes were evaluated through month 24 for patients with advanced PD with non-missing observations.
Patient population: PROSPECT enrolled 229 adults (≥30 years) with PD who had inadequately controlled motor symptoms, defined as ≥2.5 hours/day of ‘OFF’ time, despite adequate trials of available oral/transdermal/sublingual/inhalable medication. A total of 206 patients with known treatment status completed the study. A subgroup analysis of the PROSPECT study, evaluating the long-term clinical outcomes and treatment patterns in people with advanced PD whose symptoms are inadequately controlled by their current therapy, included 44 patients who were offered and initiated on a DAT as well as 185 patients who continued to receive oral medication only.
Methods: The 24-month study was conducted across several countries. Patients were assessed at months 12 and 24. For the subgroup analysis, patients were assessed at months 6, 12, 18 and 24 and outcomes were evaluated in two subgroups: those who remained on oral PD medications only, and those who initiated DAT during any point in the study. Real-world evidence is collected outside of controlled clinical trials and has inherent limitations, including a lesser ability to control for confounding factors.
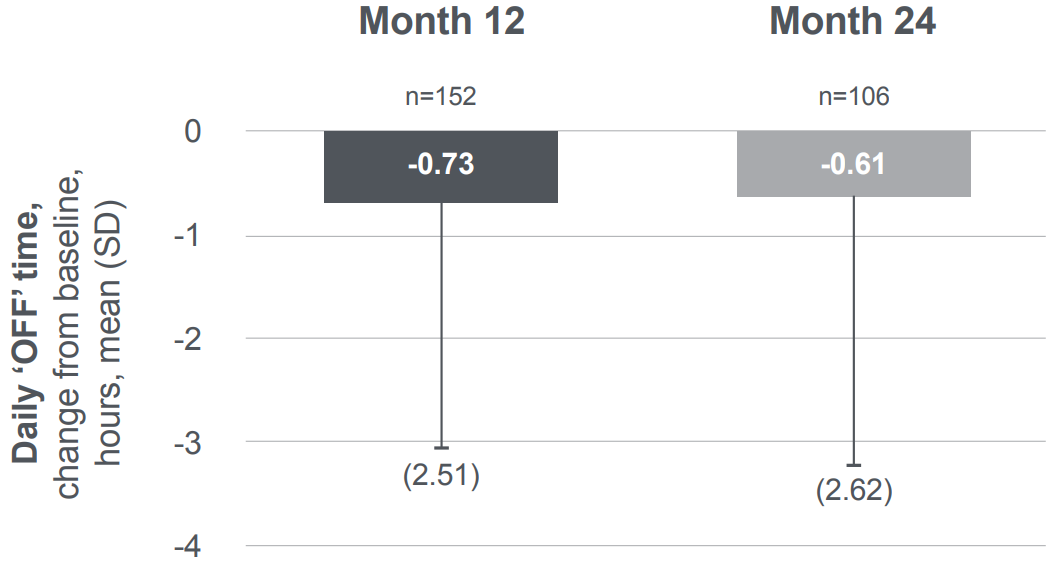
Motor symptoms remained uncontrolled for patients exclusively on oral PD medication4
As shown by the PROSPECT study, patients show clinically negligible long-term changes in daily ‘OFF’ time from baseline:4
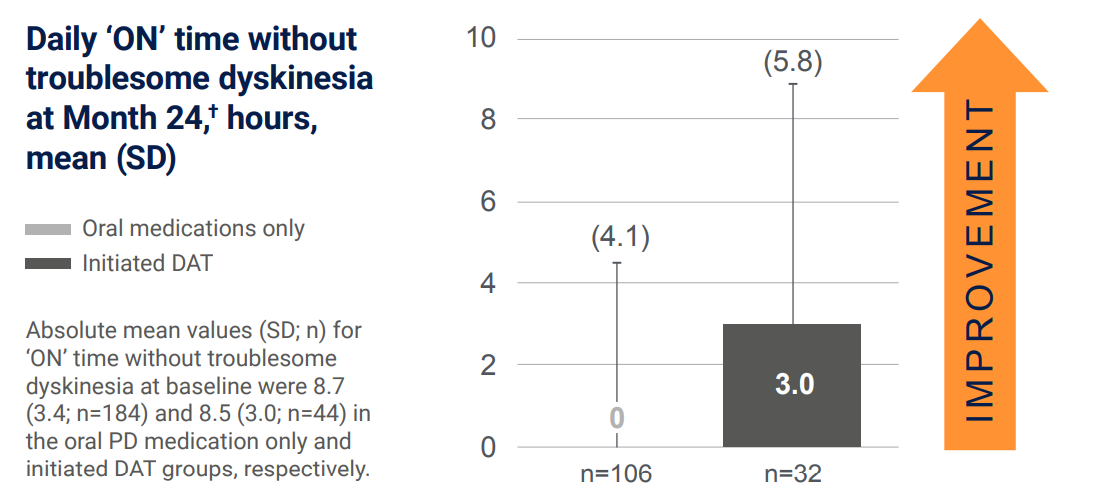
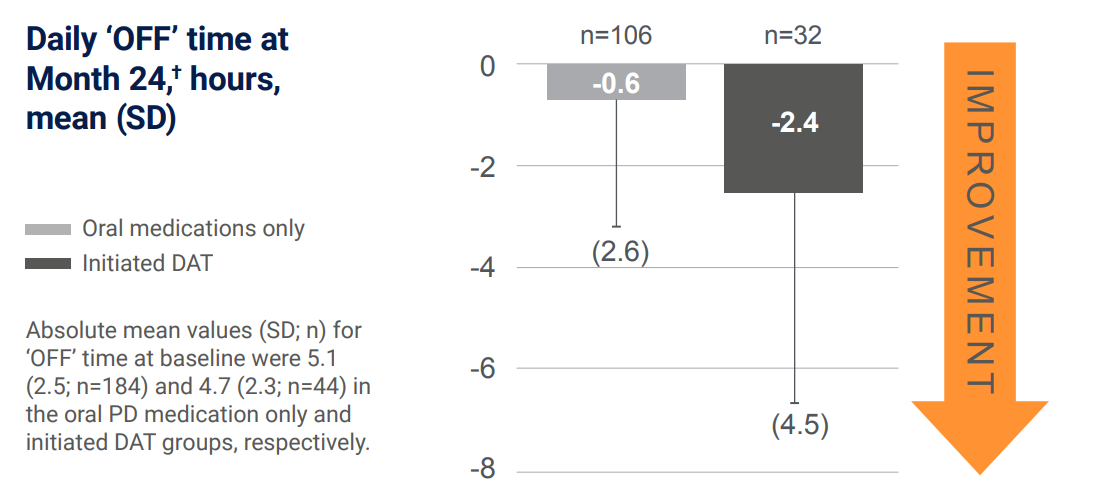
Initiation of DATs led to motor symptom improvement in advanced PD patients vs baseline5
In the sub-group analysis of the PROSPECT study, compared with people with advanced PD who continued oral medication, those who initiated DAT had greater improvement from baseline to month 24 in ‘OFF’ time and ‘ON’ time without troublesome dyskinesia:5
What might be impacting your patients’ choices around DATs?
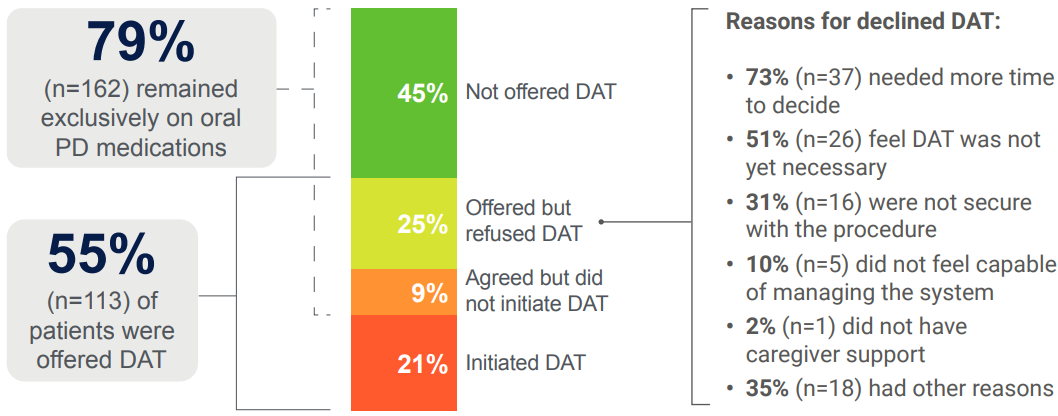
Despite the availability of DATs, 79% (162/206) of advanced PD patients in the PROSPECT study continued oral therapy alone. The most common cited reasons for those who were offered but refused a DAT are highlighted below. Of those who were offered but refused a DAT, 73% (37/51) reported they needed more time to decide.4,5
Remember James
‘My doctor has talked to me about DATs because my pill burden classifies me as having advanced PD, but I think I still need more time to decide. It’s good to know that DATs could be an option.’
This fictional case study has been developed solely for education and illustrative purposes.
Consider giving your patients the time they might need to think about their options
When should you start the conversation to give your patients the time to decide?
Remember Susan
‘My doctor has previously talked to me about the possibility of a DAT. However, because of my age, I want to first discuss with my daughter how I would manage this on a day-to-day basis and how it would fit into my everyday routine.’
This fictional case study has been developed solely for education and illustrative purposes.
Consider giving your patients the time they might need to think about their options
When should you start the conversation to give your patients the time to decide?
Remember Rahul
‘My doctor has briefly mentioned the DATs available to me; however, I still have unanswered questions about what my day-to-day routine would look like, what monitoring would involve, and whether there would be any travel limitations that might reduce my independence. I think I still need more information about what support I might need from my carer before starting therapy.’
This fictional case study has been developed solely for education and illustrative purposes.
Consider giving your patients the time they might need to think about their options
When should you start the conversation to give your patients the time to decide?
What might be the impact of advanced PD on your patients?
Supported by the 5-2-1 criteria, you can:
PRODUODOPA is indicated for the treatment of advanced levodopa-responsive PD with severe motor fluctuations and hyperkinesia or dyskinesia when available combinations of Parkinson’s medicinal products have not given satisfactory results.6
DUODOPA is indicated for the treatment of advanced levodopa-responsive PD with severe motor fluctuations and hyperkinesia or dyskinesia when available combinations of Parkinson’s medicinal products have not given satisfactory results.7
*‘OFF’ time ≥ 2.5 hours per day.
†Data was normalised to a 16-hour walking day, using the valid Hauser PD diaries completed by the person with PD 3 days before each visit. Patients could initiate DAT at any time during the study, so efficacy at the study visit may not yet have been fully established.
DAT=device-aided therapy; DBS=deep brain stimulation; PD=Parkinson’s disease; SD=standard deviation.
- Mills J, Martin A. Br J Neurosci Nurs. 2015;11(2):92–7.
- Thanvi BR, Lo TCN. Postgrad Med J. 2004;80(946):452–8.
- Mouchaileh N, et al. Aust Prescr. 2025;48(1):10–7.
- Espay A, et al. Mov Disord. 2024; 39 (suppl 1). Available from: https://www.mdsabstracts.org/abstract/long-term-evaluation-of-advanced-parkinsons-disease-burden-and-clinical-outcomes-results-from-theprospect-observational-study/. Accessed: March 2026.
- De Fabregues O, et al. Mov Disord. 2024; 39 (suppl 1). Available from: https://www.mdsabstracts.org/abstract/long-term-evolution-of-advanced-parkinsons-disease-burden-subgroup-analysis-from-the-24-month-international-prospect-observational-study/. Accessed: March 2026.
- PRODUODOPA (foslevodopa/foscarbidopa solution for infusion) UK Summary of Product Characteristics.
- DUODOPA (levodopa/carbidopa intestinal gel) UK Summary of Product Characteristics.
By clicking the links above you will leave the AbbVie Pro website and be taken to the eMC PI portal website
UK-PRODD-260089. Date of preparation: March 2026