Timing matters: Bring device-aided therapies (DATs) early into the conversation1,2
What is the real impact of PD progression?
The treatment landscape for patients with PD3,4
Levodopa remains the first line treatment option in the UK for patients in the early stages of Parkinson’s disease with motor symptoms impacting their daily life.3 For patients in the early stages whose motor symptoms do not yet impact their quality of life, initial pharmacological options may include dopamine agonists or monoamine oxidase B (MAO-B) inhibitors.3
As the disease progresses and motor fluctuations emerge, therapy is typically optimised by introducing adjunctive dopamine agonists, MAO-B inhibitors, or catechol-O-methyl transferase (COMT) inhibitors.3 If dyskinesia develops despite the optimisation of PD medications, amantadine may be considered.3
When a patient progresses to advanced PD, device-aided therapies (DATs) may be an option.4 DATs are specialised, non-oral treatments used to manage disabling motor fluctuations and dyskinesias that can not be adequately managed by oral or transdermal therapies alone.4
As PD progresses, intermittent oral therapy may no longer provide adequate control, causing decline in daily functioning5,6
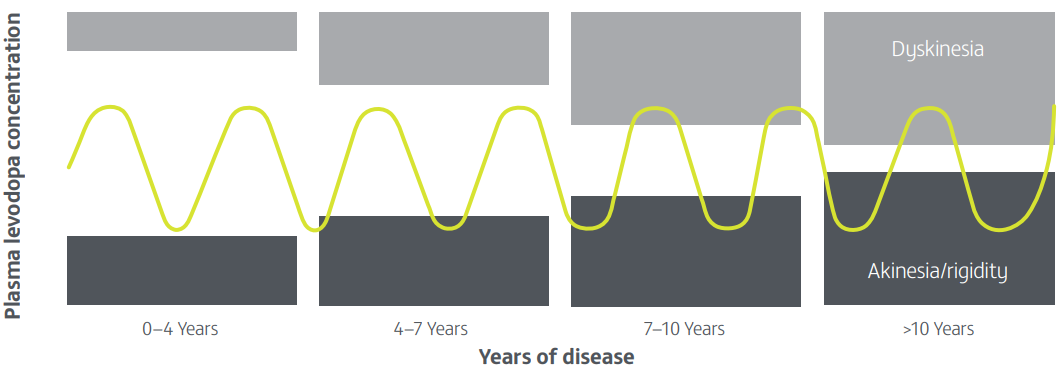
Disease progression and the accompanying narrowing of the therapeutic window10,11
As PD progresses, consistent motor control with oral levodopa-based therapy becomes harder to achieve10,11
Adapted from Cenci MA. 2014.11
For the patient, this can result in:10
- An increase in 'OFF' time
- Susceptibility to 'on' time with dyskinesia
- Negative impact on HRQoL
- Limitations in ability to function independently
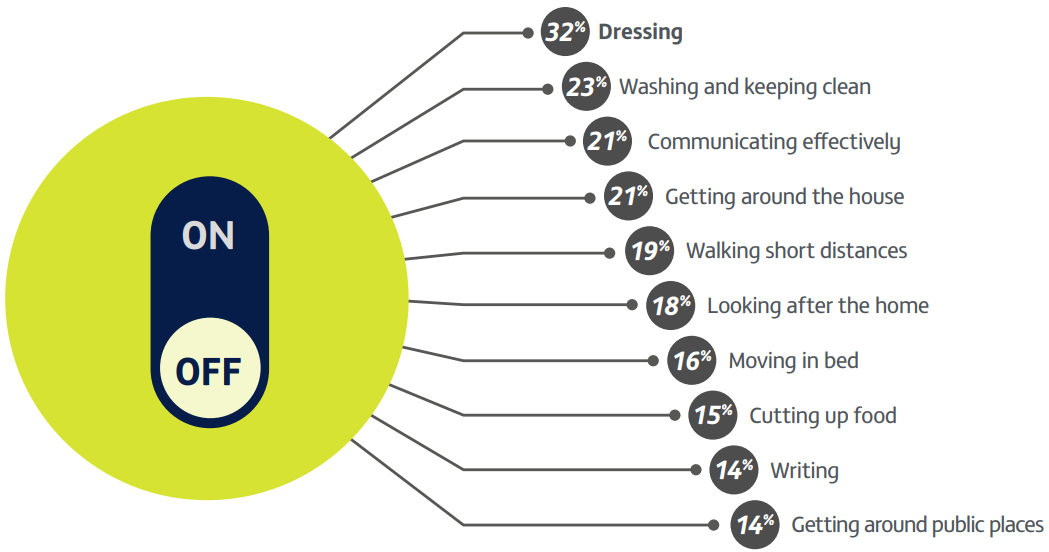
Kerr C, et al. 201612
Study type: Online survey.
Aim: To explore the impact of ‘OFF’ time on HRQoL and daily functioning in people with PD relative to ‘ON’ time.
Patient population: 305 people across the UK, France, Spain and Italy with advanced PD experiencing motor fluctuations/wearing-off with levodopa.
Methods: The survey explored: (1) the impact of ‘OFF’ time on HRQoL, (2) the impact of ‘OFF’ time on functioning and ability to undertake usual activities, and (3) the value of ‘OFF’ time relative to other factors associated with PD through a SPDCE.
Advanced PD patients may face a treatment gap14–16
When optimised oral medication regimens fail to provide adequate control for patients with advanced PD, non-oral therapies may be considered.3,4
Remember James
‘Since starting oral dopaminergic treatment seven years ago, my pill burden has become a disruptive part of my and my wife’s life.
It’s exhausting to constantly think about when I need to take each dose, and I feel like my entire day is built around my medication schedule. It makes it harder to keep my Parkinson’s diagnosis at the back of my mind. Planning meals out with friends can be especially challenging because I’m always trying to coordinate the timing of my pills. I also often have to wake up earlier than I’d like to make sure I stay on schedule.’
This fictional case study has been developed solely for education and illustrative purposes.
Start the conversation about non-oral therapies with patients like James early in the disease course1,17
Remember Susan
‘I’ve been taking oral dopaminergic medication for over five years, with the dose schedule increasing over time. Still, I suffer from periods where I slow down, stiffen up, and even simple tasks around the house become difficult. This can add up to more than 2 hours of my day. My doctor called this disruptive OFF time.
What troubles me most is not knowing when an ‘OFF’ episode will strike. It makes it hard to plan time with my family—especially with my young grandchild.’
This fictional case study has been developed solely for education and illustrative purposes.
Start the conversation about non-oral therapies with patients like Susan early in the disease course1,17
Remember Rahul
‘I’ve been taking oral dopaminergic medication since I was diagnosed eight years ago, and over time I’ve needed to take it more and more often. Even so, I still suffer from periods where I’m unable to control my arms or legs, which my doctor calls dyskinesia. This can disturb at least an hour of my day, is unpredictable, and can be frightening—especially when it makes simple things like eating or getting dressed difficult.
I value my independence and, even though I have a part-time carer, the thought of having a dyskinesia episode while I’m alone at home makes me anxious. Also, because the movements are so visible, I often avoid meeting people in public for fear it will happen in front of them.’
This fictional case study has been developed solely for education and illustrative purposes.
Start the conversation about non-oral therapies with patients like Rahul early in the disease course1,17
What might be the impact of advanced PD on your patients?
Supported by the 5-2-1 criteria, you can:
PRODUODOPA is indicated for the treatment of advanced levodopa-responsive PD with severe motor fluctuations and hyperkinesia or dyskinesia when available combinations of Parkinson’s medicinal products have not given satisfactory results.18
DUODOPA is indicated for the treatment of advanced levodopa-responsive PD with severe motor fluctuations and hyperkinesia or dyskinesia when available combinations of Parkinson’s medicinal products have not given satisfactory results.19
DAT=device-aided therapy; HRQoL=health-related quality of life; PD=Parkinson’s disease; SPDCE=stated preference discrete choice experiment.
- Mills J, Martin A. Br J Neurosci Nurs. 2015;11(2):92–7.
- Thanvi BR, Lo TCN. Postgrad Med J. 2004;80(946):452–8.
- NICE. Parkinson’s disease in adults. Available at: www.nice.org.uk/guidance/ng71. Accessed: March 2026.
- Mouchaileh N, Cameron J. Aust Prescr. 2025;48:10-17.
- Dijk JM, et al. J Parkinsons Dis. 2020;10:S65–73.
- Varanese S, et al. Parkinsons Dis. 2011;2010:480260.
- Doi H, et al. J Neurol Sci. 2012;319:86–8.
- Hardoff R, et al. Mov Disord. 2001;16:1041–7.
- Nyholm D. Parkinsonism Relat Disord. 2007;13(suppl):S13–S7.
- Lundqvist C. Neuropsychiatr Dis Treat. 2007;3(3):335–48.
- Cenci MA. Front Neurol. 2014;5(242):1–15.
- Kerr C, et al. Qual Life Res. 2016;25(6):1505–15.
- Rastgardani T, et al. Front. Neurol. 2019;10:892. doi: 10.3389/fneur.2019.00892.
- Parkinson’s UK. 2019 Parkinson’s audit: Summary report. Available from: https://www.parkinsons.org.uk/sites/default/files/2020-01/CS3524%20Parkinson%27s%20UK%20Audit%20-%20Summary%20Report%202019%20%281%29.pdf. Accessed: March 2026.
- AbbVie. Data on file, HPA data for DAT patients, UK-DUOD-240036.
- Parkinson’s UK. Parkinson’s diagnoses on the rise in the UK. Available at: http://parkinsons.org.uk/professionals/news/2018/parkinsons-diagnoses-rise-uk#. Accessed March 2026.
- Worth PF. Pract Neurol 2013; 13:140–52.
- PRODUODOPA (foslevodopa/foscarbidopa solution for infusion) UK Summary of Product Characteristics.
- DUODOPA (levodopa/carbidopa intestinal gel) UK Summary of Product Characteristics.
By clicking the links above you will leave the AbbVie Pro website and be taken to the eMC PI portal website
UK-PRODD-260088. Date of preparation: March 2026