Timing matters: Bring device-aided therapies (DATs) early into the conversation1,2
Is it time to review therapy?
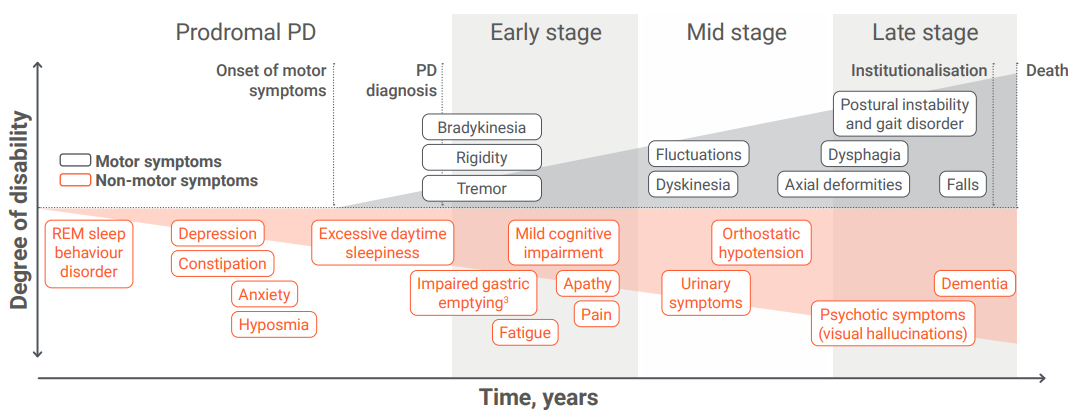
At what stage would you diagnose advanced PD?
Adapted from Poewe W, et al. 2017.3,4
PD is a progressive neurodegenerative disorder, with symptom management becoming more complex over time.4 There is no simple way to diagnose advanced PD, and the concept of advanced disease5—along with the suitability of patients for advanced, non-oral therapies5—is not defined by a specific point on the disease timeline, but rather by increasing symptom complexity and a significant impact on daily life.6 These challenges may make it difficult to recognise advanced PD.7
5-2-1 criteria: A tool designed to identify patients with advanced PD5
While determining whether a patient has reached advanced PD can be difficult due to the lack of biomarkers and the varied nature of PD symptoms, the 5-2-1 criteria offer a tool to identify possible transition to advanced disease.5
The 5-2-1 criteria were created in an AbbVie-funded study by a modified Delphi panel approach, where an iterative, structured consensus process was used for eliciting and refining the opinions of a group of experts.5
Meeting any ONE of the following criteria may suggest a patient is transitioning into the advanced stage of PD and could be considered for non-oral therapy:5
Meet James
A 62-year-old editor living with his wife
Since embarking on an oral dopaminergic treatment seven years ago, his pill burden has become a disruptive aspect of his and his wife’s life.
James currently takes 100 mg/25 mg levodopa/carbidopa sustained release five times a day.
James fits the 5-2-1 criteria for advanced PD.5
This fictional case study has been developed solely for education and illustrative purposes.
Could some of your patients, like James, already be in the advanced stage of PD?
Meet Susan
A 69-year-old part-time tutor who recently became a grandmother
She has been treated with an oral dopaminergic for over five years.
Despite gradually increased frequency of dosing, Susan experiences disruptive and distressing ‘OFF’ time for over two hours every day.
Susan fits the 5-2-1 criteria for advanced PD.5
This fictional case study has been developed solely for education and illustrative purposes.
Could some of your patients, like Susan, already be in the advanced stage of PD?
Meet Rahul
A 66-year-old retired piano player who lives alone but has a part-time carer
He has been taking an oral dopaminergic since his diagnosis eight years ago.
The frequency of his dosing has steadily climbed, he suffers from disabling dyskinesia for at least an hour every day.
Rahul fits the 5-2-1 criteria for advanced PD.5
This fictional case study has been developed solely for education and illustrative purposes.
Could some of your patients, like Rahul, already be in the advanced stage of PD?
Antonini A, et al. 20185
Study type: Delphi consensus.
Aim: To reach consensus among movement disorder specialists regarding key patient characteristics indicating transition to advanced PD and guiding appropriate use of non-oral therapies* in the management of PD.
Panellists: Leading movement disorder specialists from 10 EU countries were recruited. 17 panellists were chosen based on their recognised clinical expertise treating PD, breadth and depth of advanced PD research, as well as their experience with the use of non-oral therapies* for the treatment of advanced PD.
Methods: A modified Delphi panel approach was utilised. The Delphi process has been widely used for achieving convergence of opinion from a panel of experts.
*Non-oral therapy=device-aided therapy.
What might be the impact of advanced PD on your patients?
Supported by the 5-2-1 criteria, you can:
PRODUODOPA is indicated for the treatment of advanced levodopa-responsive PD with severe motor fluctuations and hyperkinesia or dyskinesia when available combinations of Parkinson’s medicinal products have not given satisfactory results.9
DUODOPA is indicated for the treatment of advanced levodopa-responsive PD with severe motor fluctuations and hyperkinesia or dyskinesia when available combinations of Parkinson’s medicinal products have not given satisfactory results.10
DAT=device-aided therapy; PD=Parkinson’s disease; REM=rapid eye movement.
- Mills J, Martin A. Br J Neurosci Nurs. 2015;11(2):92–7.
- Thanvi BR, Lo TCN. Postgrad Med J. 2004;80(946):452–8.
- Fasano A, et al. Lancet Neurol. 2015;14:625–35.
- Poewe W, et al. Nat Rev Dis Primers. 2017;3:17013.
- Antonini A, et al. Curr Med Res Opin. 2018;34:2063–73.
- Parkinson’s UK. Advanced Parkinson’s. Available from: https://www.parkinsons.org.uk/information/about-parkinsons/advanced. Accessed: March 2026.
- Fasano A, et al. Acta Neurol Scand. 2022;146:167–176.
- Parkinson’s UK. 2025 Parkinson’s audit: Summary report. Available from: https://www.parkinsons.org.uk/sites/default/files/2026-02/244549%20-%20Transforming%20care_%20report%20of%20the%202025%20UK%20Parkinson_s%20Audit.pdf. Accessed: March 2026.9.
- PRODUODOPA (foslevodopa/foscarbidopa solution for infusion) UK Summary of Product Characteristics
- DUODOPA (levodopa/carbidopa intestinal gel) UK Summary of Product Characteristics.
By clicking the links above you will leave the AbbVie Pro website and be taken to the eMC PI portal website
UK-PRODD-260087. Date of preparation: March 2026