*Oral tablet.
†Administer intravenously. Dose may be split as 100 mg and 900 mg on Days 1 and 2 of Cycle 1, respectively. Graphic not to scale. Each cycle is 28 days.
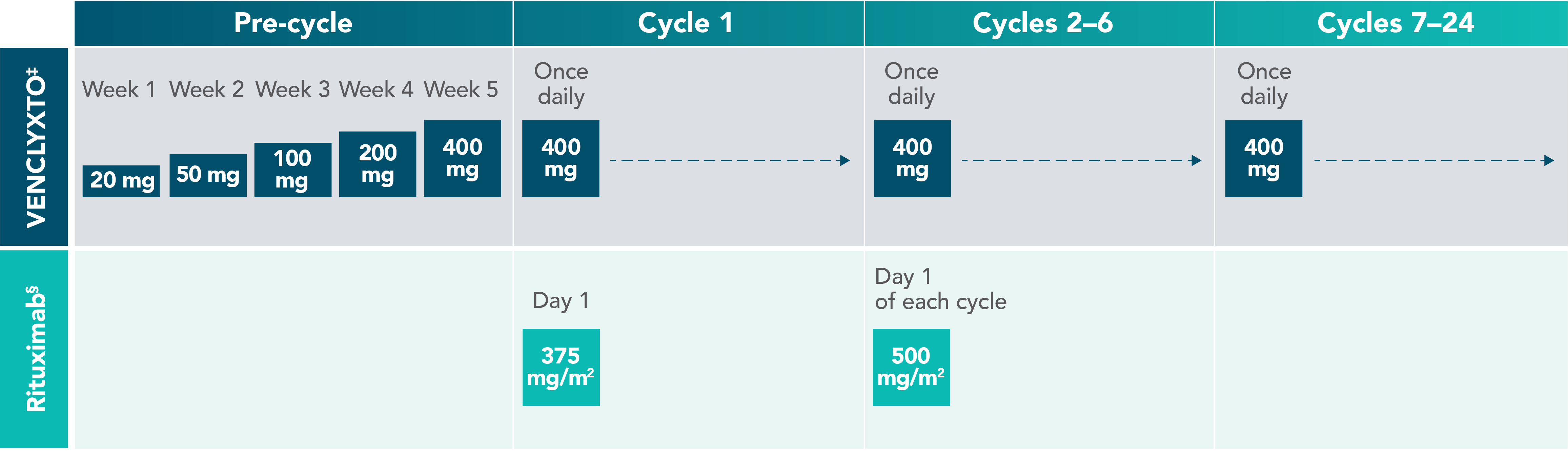
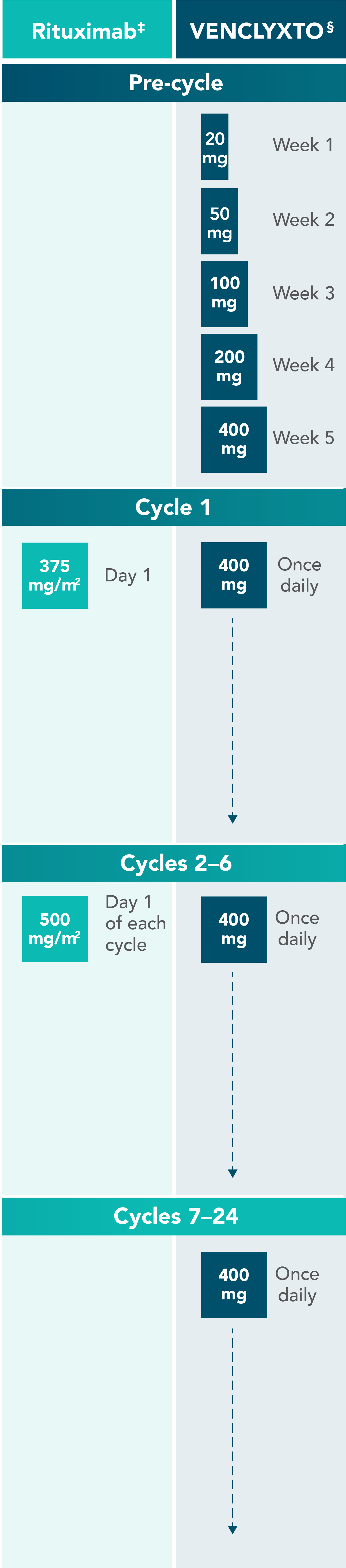
VENCLYXTO® + rituximab: Designed to be completed in ~2 years in 2L+ CLL1
‡Oral tablet.
§Administer intravenously.
Graphic not to scale. Each cycle is 28 days.
‡Administer intravenously.
§Oral tablet.
Graphic not to scale. Each cycle is 28 days.
CLL=chronic lymphocytic leukaemia; 2L+=second line + later lines of therapy; HR=hazard ratio; CI=confidence interval; PFS=progression-free survival; BR=bendamustine + rituximab; VEN+R=VENCLYXTO® + rituximab; TLS=tumour lysis syndrome.
▼This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions via HPRA. Website: www.hpra.ie.
I want to find out more
about VENCLYXTO®
I want to receive more information
about VENCLYXTO®
Reference: 1. VENCLYXTO® Summary of Product Characteristics, available at www.medicines.ie 2. Kater AP, Kipps TJ, Eichhorst B, et al. Five-year analysis of MURANO study demonstrates enduring undetectable minimal residual disease (uMRD) in a subset of relapsed/refractory chronic lymphocytic leukemia patients following fixed-duration venetoclax-rituximab therapy. Oral presentation (125) presented at: 62nd American Society of Hematology Annual Meeting and Exposition; December 5-8, 2020; Virtual meeting
IE-VNCCLL-200097 | Date of preparation: December 2020