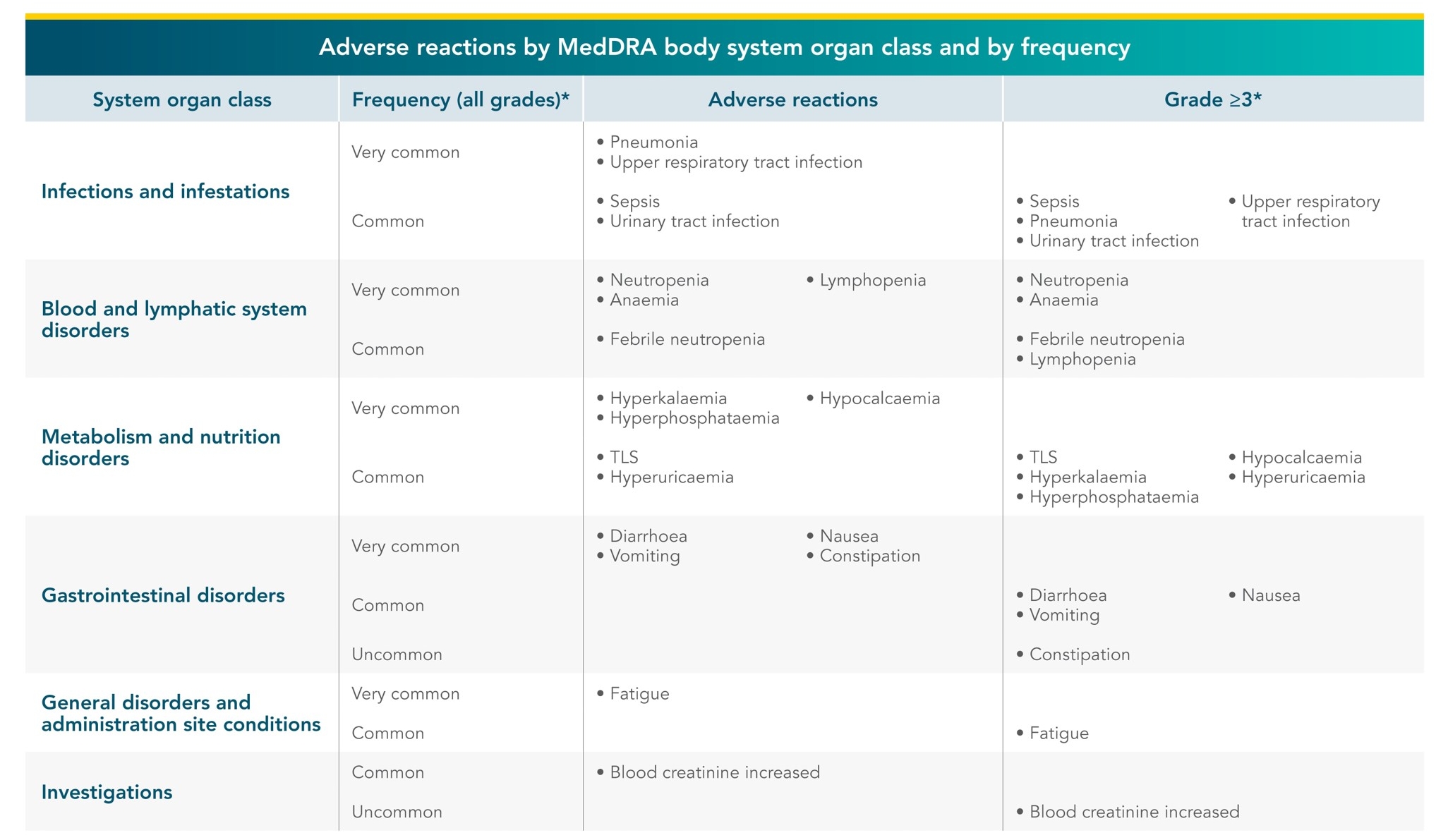
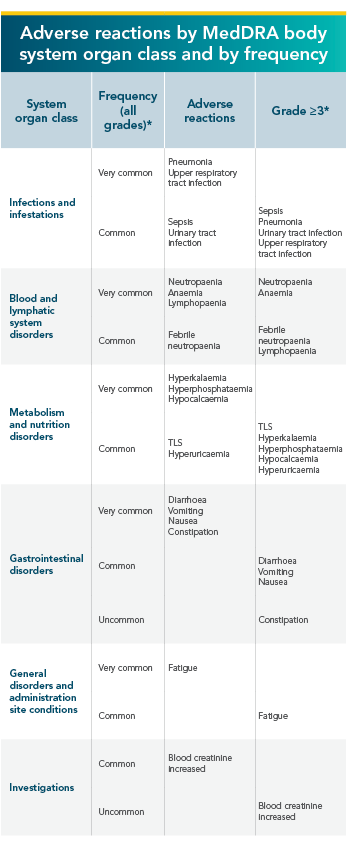
VENCLYXTO®-based regimens have an established and manageable safety profile in CLL clinical trials1
- The most frequently reported serious adverse reactions (≥2%) in patients receiving VENCLYXTO® in combination with rituximab were pneumonia, sepsis, febrile neutropenia, and TLS. Serious infections including events of sepsis with fatal outcome have been reported. Patients require monitoring for signs and symptoms of infection and promt treatment.
- The most commonly occurring adverse reactions (≥20%) of any grade in patients receiving VENCLYXTO® in the combination with rituximab were neutropenia, diarrhoea, and upper respiratory tract infection
*Only the highest frequency observed in the trials is reported (based on studies CLL14, MURANO, M13-982, M14-032, and M12-175).
TLS=tumour lysis syndrome; MedDRA=Medical Dictionary for Regulatory Activities
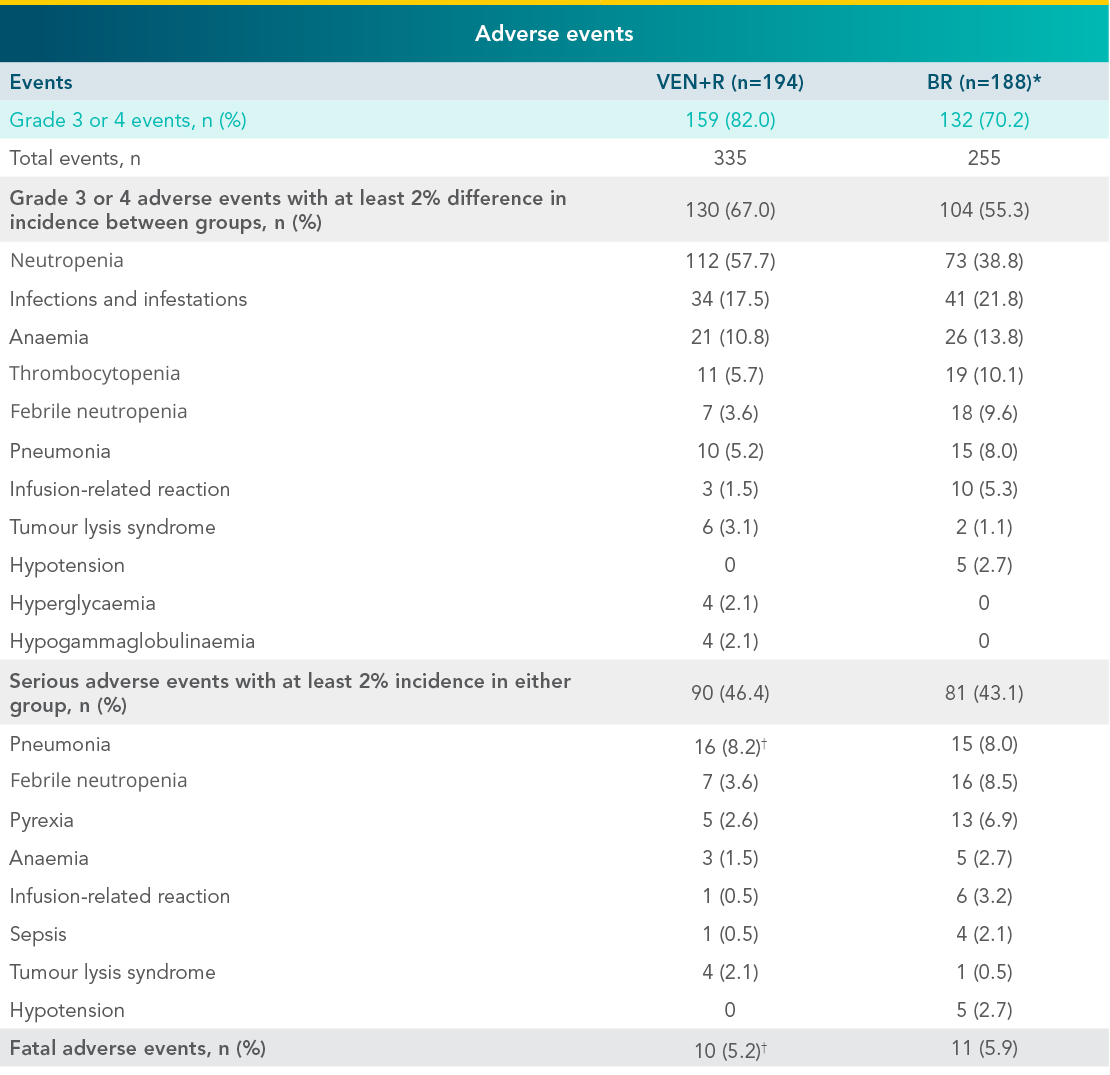
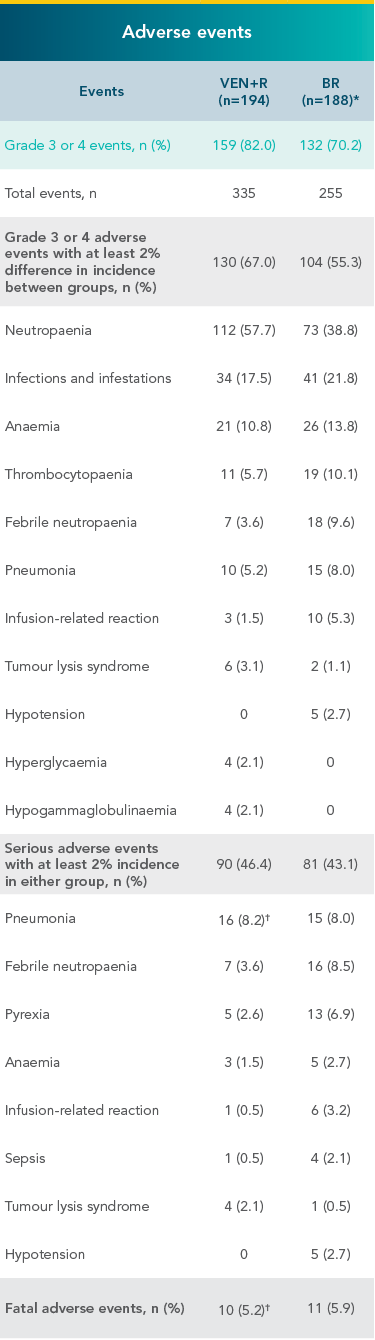
Grade 3 or 4 adverse events in the MURANO VEN+R and BR treatment arms3
- The duration of treatment was longer in the VEN+R arm than in the BR arm
*7 patients in the BR arm who did not receive treatment were not included in the safety analysis.
†2 serious adverse events of pneumonia that resulted in death occurred in patients who had both disease progression and confirmed Richter’s transformation (ie, conversion into an aggressive lymphoma, typically diffuse large B-cell lymphoma).
CLL=chronic lymphocytic leukaemia; 2L+=second line + later lines of therapy; HR=hazard ratio; CI=confidence interval; PFS=progression-free survival; BR=bendamustine + rituximab; VEN+R=VENCLYXTO® + rituximab; TLS=tumour lysis syndrome; MedDRA=Medical Dictionary for Regulatory Activities;
▼This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions via HPRA. Website: www.hpra.ie.
I want to find out more
about VENCLYXTO® in CLL
I want to receive more information
about VENCLYXTO® in CLL
References: 1. VENCLYXTO® Summary of Product Characteristics, available at www.medicines.ie 2. Kater AP, Kipps TJ, Eichhorst B, et al. Five-year analysis of MURANO study demonstrates enduring undetectable minimal residual disease (uMRD) in a subset of relapsed/refractory chronic lymphocytic leukemia patients following fixed-duration venetoclax-rituximab therapy. Oral presentation (125) presented at: 62nd American Society of Hematology Annual Meeting and Exposition; December 5-8, 2020; Virtual meeting 3. Seymour JF, Kipps TJ, Eichhorst B, et al. Venetoclax–rituximab in relapsed or refractory chronic lymphocytic leukemia. N Engl J Med. 2018;378(12):1107‑1120.
IE-VNCCLL-200105 | Date of preparation: December 2020