UK-RISN-230338. Date of preparation January 2024.
Find out more about the efficacy of SKYRIZI
Explore the durability of response with SKYRIZI through clinical trials and long-term data in psoriasis and psoriatic arthritis. Start by selecting a study from the menu below.
LONG-TERM DATA
HEAD-TO-HEAD TRIALS
SKYRIZI IN PsA
HEAD-TO-HEAD TRIALS: SKYRIZI vs Secukinumab (IMMerge)
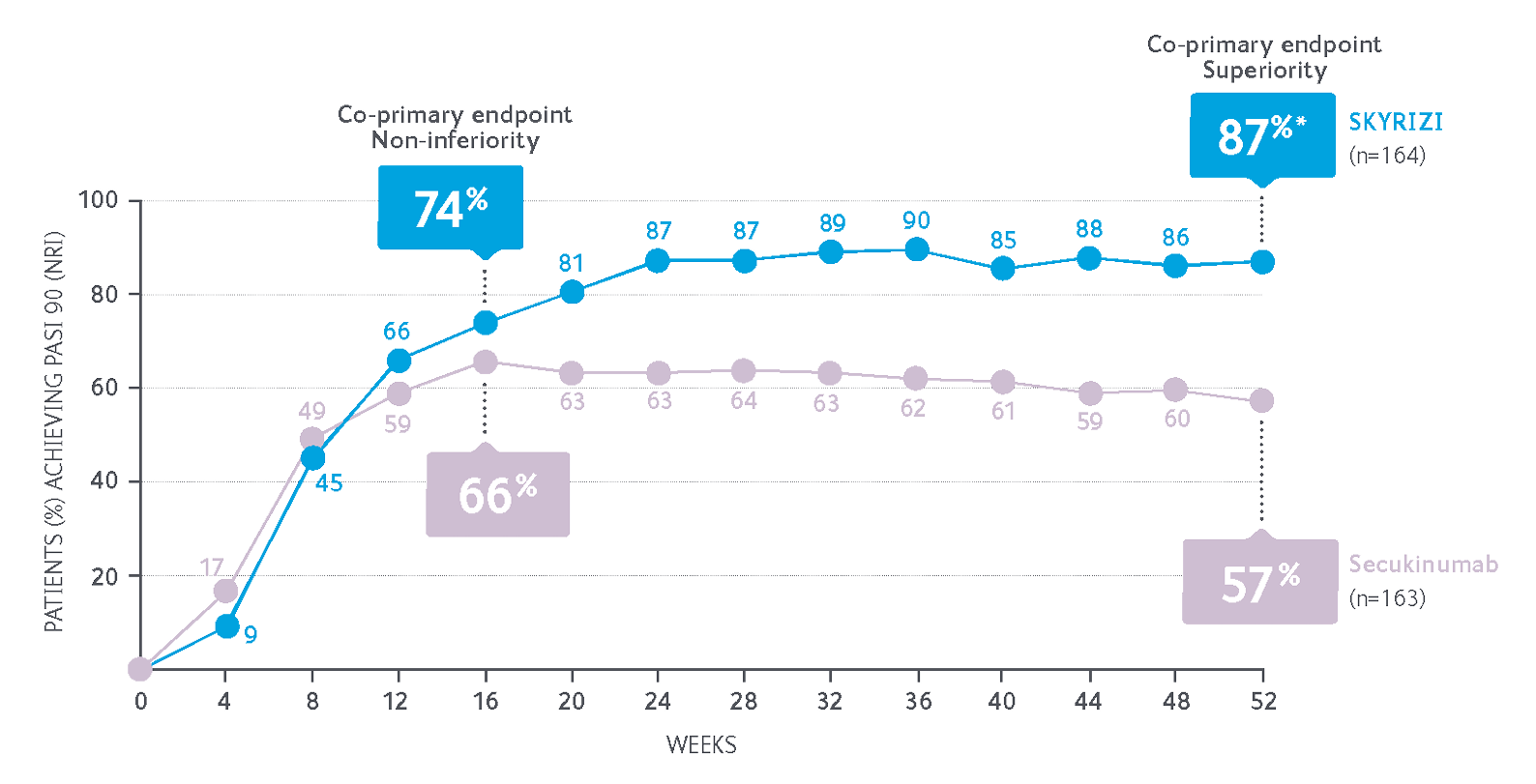
Superior efficacy in psoriasis with fewer injections vs secukinumab at 52 Weeks3,5
87% of SKYRIZI patients achieved PASI 90 at Week 52 after 5 doses compared with 57% of secukinumab patients after 16 doses†3,5
Adapted from Warren RB, et al. 2020.
Data from IMMerge, a Phase III, global, multicentre, randomised, open-label, efficacy assessor-blinded active-comparator study in adult patients with moderate to severe plaque psoriasis.
NRI, non-responder imputation; PASI, Psoriasis Area and Severity Index. P-values for comparison vs secukinumab: *P<0.001 multiplicity controlled.
Data assessed for intent-to-treat population. Adjusted difference CI values: 96.25% at Week 16 and 95%; P values calculated from the Cochran-Mantel-Haenszel test, stratified by weight (≤100 kg vs >100 kg) and prior systemic biologic use for psoriasis.
†Each dose for SKYRIZI given as two 75-mg subcutaneous injections, each dose for secukinumab given as two 150-mg subcutaneous injections.
SKYRIZI demonstrated superiority at Week 52 across primary and all ranked secondary endpoints.
Co-primary endpoints were met:
- Non-inferiority of SKYRIZI vs secukinumab at Week 16 (PASI 90: 74% vs 66%)
- Superiority of SKYRIZI vs secukinumab at Week 52 (PASI 90: 87% vs 57%, P<0.001)
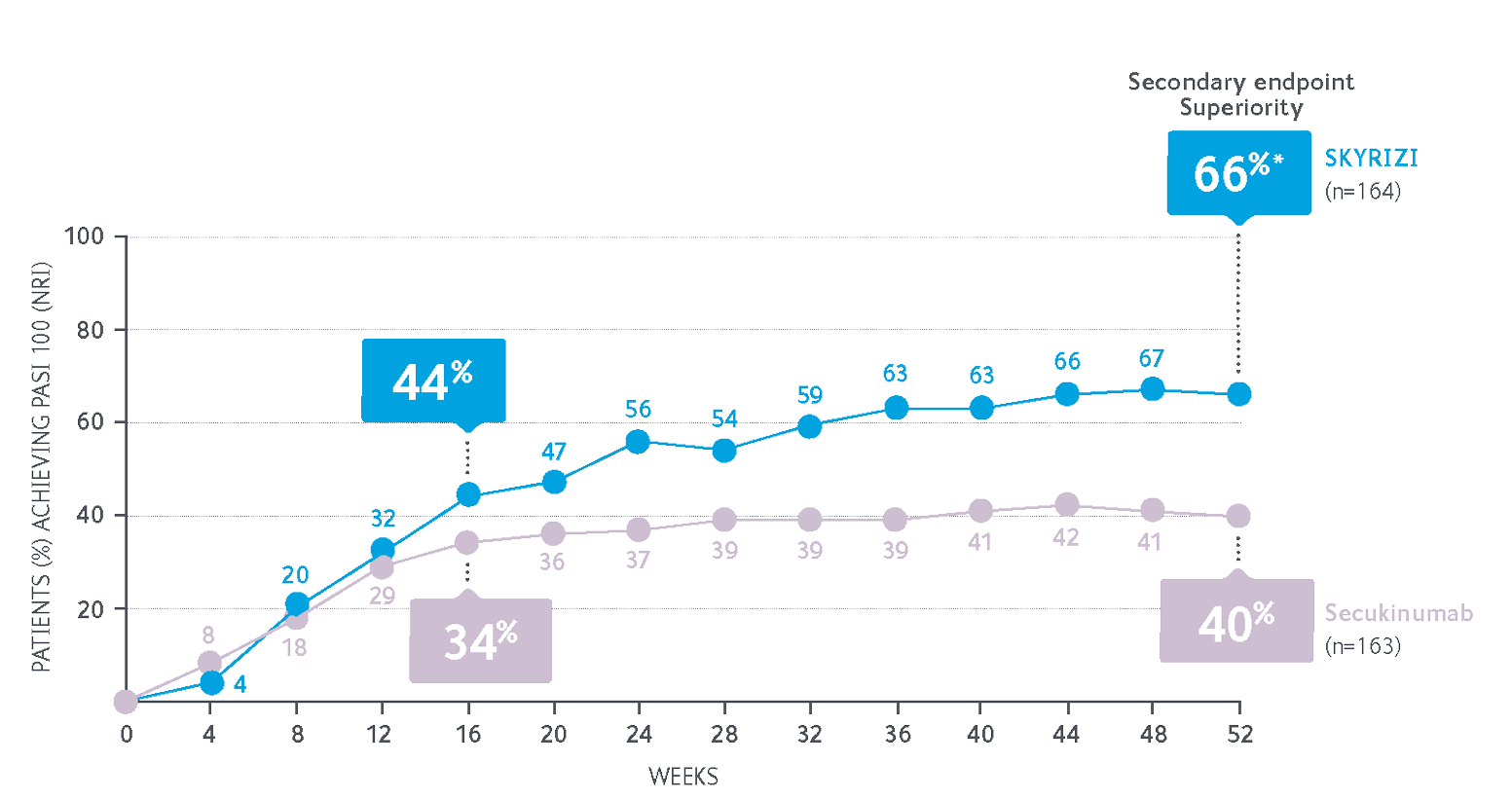
66% of SKYRIZI patients achieved PASI 100 at Week 52 after 5 doses compared with 40% of secukinumab patients after 16 doses†3,5
Adapted from Warren RB, et al. 2020.
Data from IMMerge, a phase III, global, multicentre, randomised, open-label, efficacy assessor-blinded active-comparator study in adult patients with moderate to severe plaque psoriasis.
NRI, non-responder imputation; PASI, Psoriasis Area and Severity Index. P-values for comparison vs secukinumab: *P<0.001 multiplicity controlled.
Data assessed for intent-to-treat population. Adjusted difference CI set at 95%; P values calculated from the Cochran-Mantel-Haenszel test, stratified by weight (≤100 kg vs >100 kg) and prior systemic biologic use for psoriasis.
†Each dose for SKYRIZI given as two 75-mg subcutaneous injections, each dose for secukinumab given as two 150-mg subcutaneous injections.
All ranked secondary endpoints were met, including statistically significant superiority of SKYRIZI vs secukinumab at Week 52 (PASI 100: 66% vs 40%, P<0.001). Co-primary endpoints were met: PASI 90 at Week 16 (non-inferiority) and PASI 90 at Week 52 (superiority).
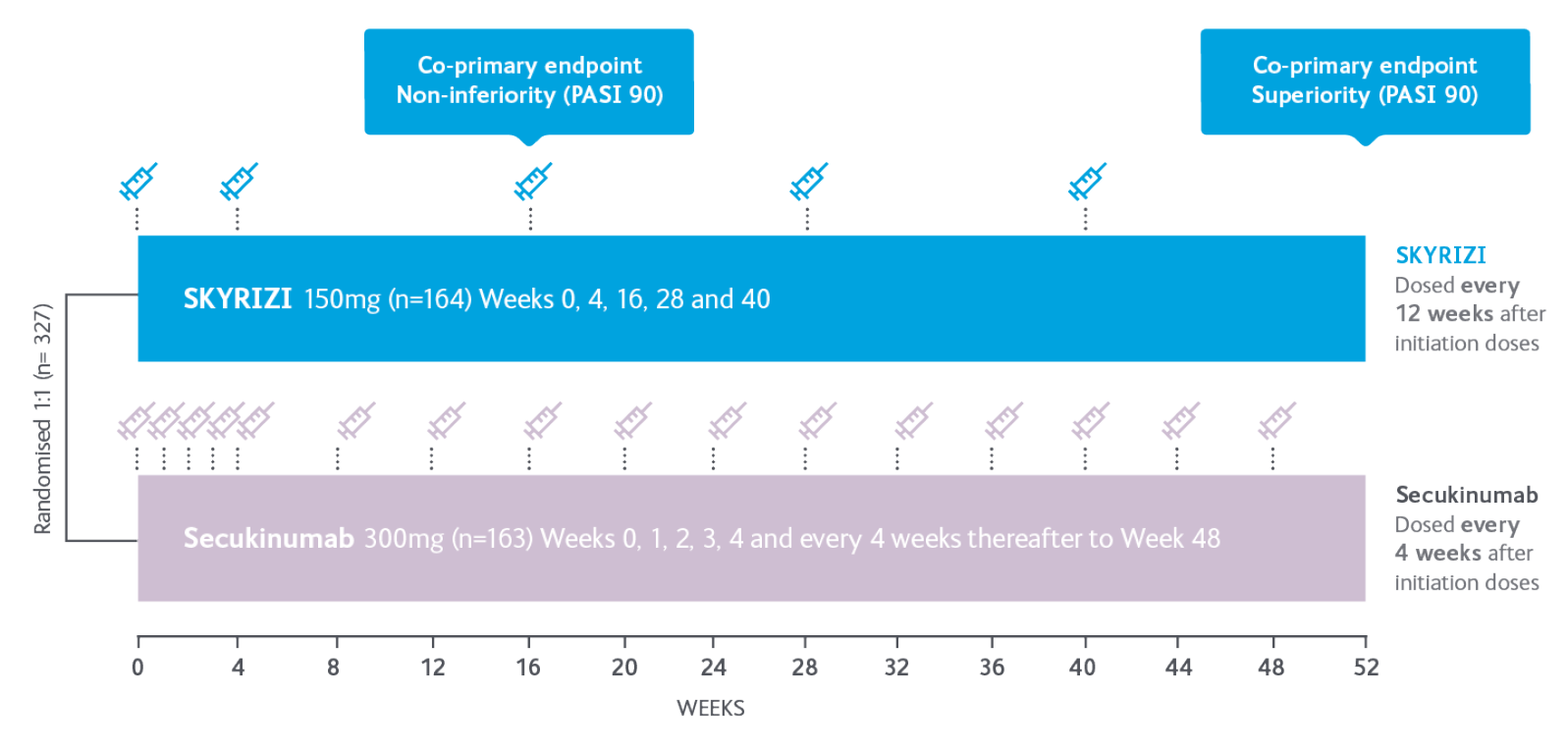
IMMerge Study Design
A Phase III, global, multicentre, randomised, open-label, efficacy assessor-blinded, active comparator study to evaluate the safety and efficacy of risankizumab vs secukinumab in adults with moderate to severe plaque psoriasis.
Adapted from Warren RB, et al. 2020.
A total of 327 subjects were randomised 1:1 to SKYRIZI (n=164) (150 mg), given as two 75-mg subcutaneous injections at baseline, 4 weeks later, and every 12 weeks thereafter, or secukinumab (n=163) (300 mg) given as two 150-mg subcutaneous injections, at baseline, weeks 1, 2, 3, and 4, and then every 4 weeks thereafter.
PASI, Psoriasis Area and Severity Index.
Co-primary endpoints
- PASI 90 at Week 52 (superiority): Proportion of patients who achieved PASI 90 response at Week 52 vs secukinumab.
- PASI 90 at Week 16 (non-inferiority): Proportion of patients who achieved PASI 90 response at Week 16 vs secukinumab.
Co-primary endpoints were met.
Ranked secondary endpoints
- PASI 100 at Week 52 vs secukinumab
- PASI 75 at Week 52 vs secukinumab
- sPGA 0/1 at Week 52 vs secukinumab
All ranked secondary endpoints were met.
PASI, Psoriasis Severity Index; sPGA, static Physicians’ Global Assessment.
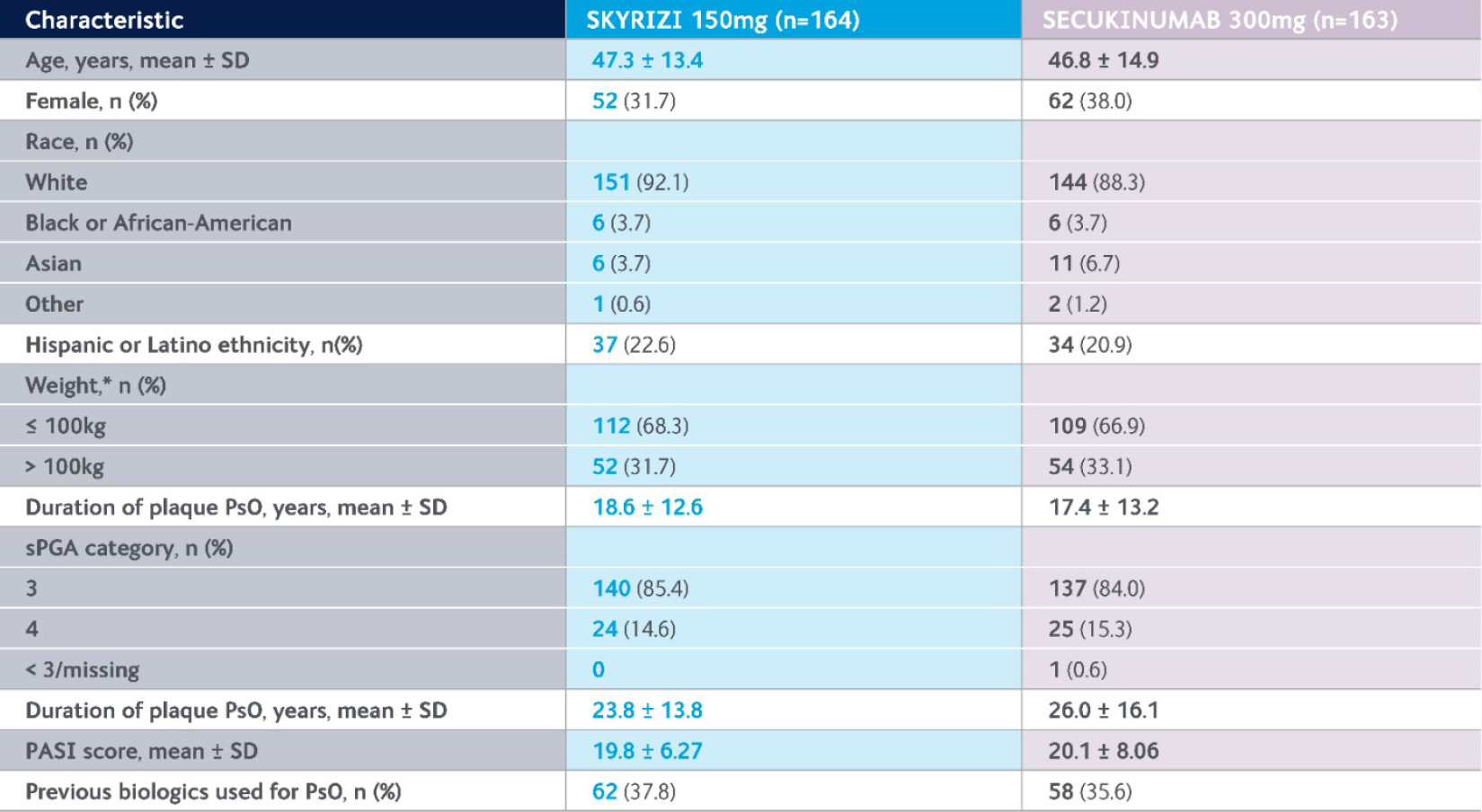
IMMerge baseline demographics3,5
Key demographics and baseline characteristics were generally balanced between treatment groups and consistent with the pivotal UltIMMa-1 and UltIMMa-2 trials.3
Adapted from Warren RB, et al. 2020.
SD, standard deviation; sPGA, static Physician Global Assessment; PASI, Psoriasis and Severity Index; PsO, psoriasis.
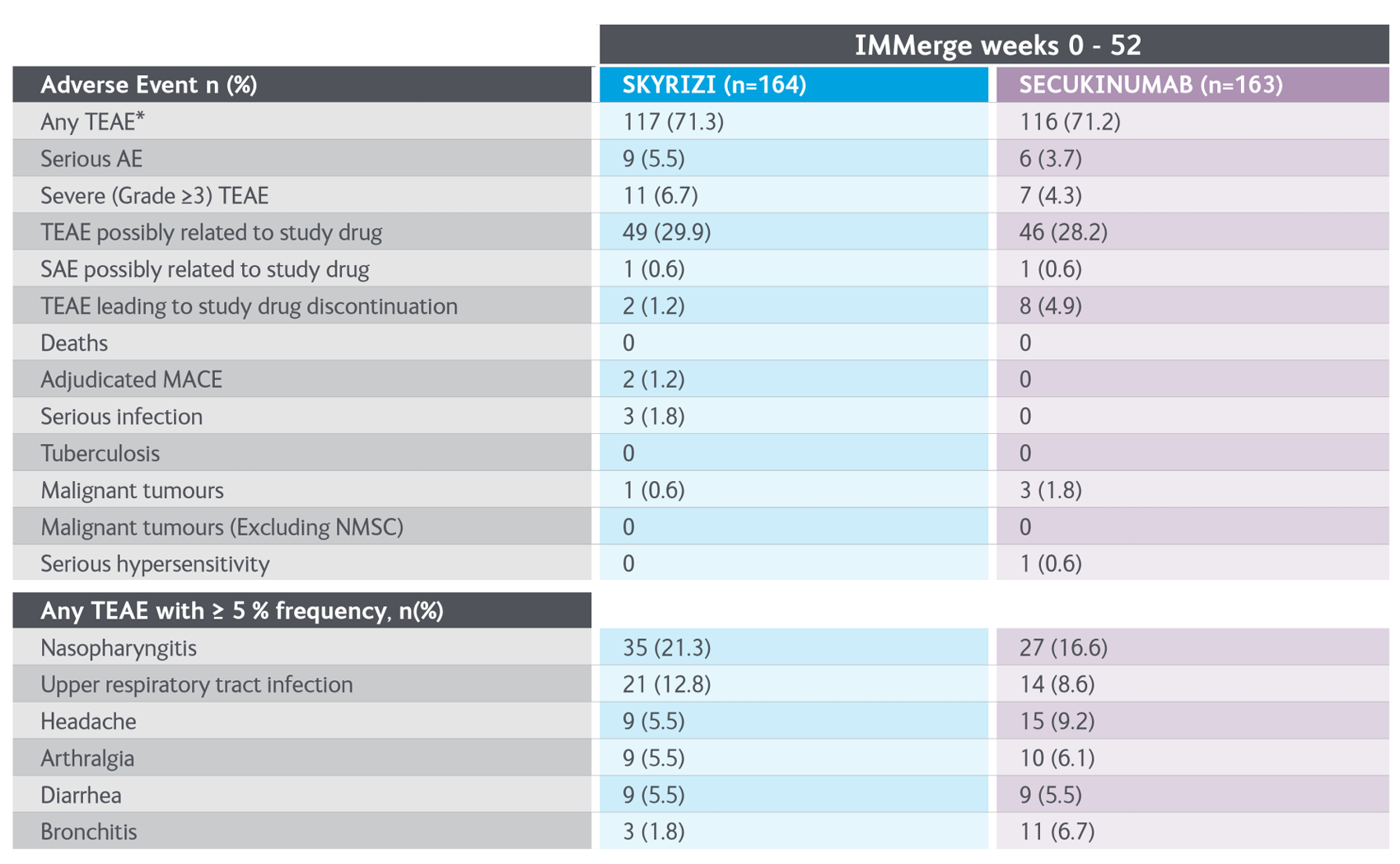
Consistent safety profile through to 52 Weeks in the IMMerge trial3
Adapted from Warren RB, et al. 2020.
*Defined as AEs occurring with an onset within 20 weeks after the last dose of study drug administration.
AE, adverse event; MACE, major adverse cardiovascular event, NMSC, non-melanoma skin cancer; TEAEs, treatment-emergent adverse events.
Featured content
UK-RISN-230313. Date of preparation February 2024.
References
- Reich K, et al. Lancet 2019; 394: 576-586.
- Gordon KB, et al. Lancet 2018; 392: 650-661.
- Warren RB, et al. Risankizumab vs Secukinumab in Patients with Moderate-to-Severe Plaque Psoriasis: A Phase 3 Trial, Presented at AAD 2020.
- SKYRIZI: Summary of Product Characteristics.
- Warren RB, et al. Br J Dermatol 2021; 185: 50-59.
UK-RISN-230312. Date of preparation: February 2024.
Important safety information for SKYRIZI®▼(risankizumab) in Psoriasis and Psoriatic Arthritis¹
SKYRIZI should be initiated and supervised by healthcare professionals experienced in the diagnosis and treatment of Psoriasis and Psoriatic arthritis.
Some patients may not be suitable for SKYRIZI (risankizumab). You are strongly advised to read the prescribing information, which can be found at the top of this webpage and below, and the Summary of Product Characteristics (SmPC) which are available online in the Electronic Medicines Compendium (EMC) from the links below.
SKYRIZI 150 mg solution for injection in pre-filled pen CLICK HERE.
SKYRIZI 150 mg solution for injection in pre-filled syringe CLICK HERE.
SKYRIZI PRESCRIBING INFORMATION CLICK HERE.
SKYRIZI is contraindicated in patients;
- with hypersensitivity to the active substance or to any of the excipients,
- with clinically important active infections (e.g. active tuberculosis).
Cautions (See Prescribing Information and SmPC for full details including screening and monitoring requirements):
It is preferable to avoid the use of SKYRIZI during pregnancy. Women of childbearing potential should use an effective method of contraception during treatment and for at least 21 weeks after treatment. It is unknown whether risankizumab is excreted in human milk. A decision should be made whether to discontinue/abstain from risankizumab therapy, taking into account the benefit of breast-feeding to the child and the benefit of risankizumab therapy to the woman.
Dosing in Psoriasis and Psoriatic Arthritis
The recommended dose of SKYRIZI is 150mg administered by subcutaneous injection at Week 0, Week 4, and every 12 weeks thereafter.
Adverse reactions
For adverse reactions, please refer to the prescribing information and the SKYRIZI summary of product characteristics available online in the Electronic Medicines Compendium via the links above.
Important safety information for HUMIRA® (adalimumab) in Psoriasis and Psoriatic Arthritis²
HUMIRA is intended for use under the guidance and supervision of a physician experienced in the diagnosis and treatment of conditions for which HUMIRA is indicated.
Psoriatic arthritis: HUMIRA is indicated for the treatment of active and progressive psoriatic arthritis in adults when the response to previous disease-modifying antirheumatic drug therapy has been inadequate. HUMIRA has been shown to reduce the rate of progression of peripheral joint damage as measured by X-ray in patients with polyarticular symmetrical subtypes of the disease and to improve physical function.
Psoriasis: HUMIRA is indicated for the treatment of moderate-to-severe chronic plaque psoriasis in adult patients who are candidates for systematic therapy
Paediatric plaque psoriasis: HUMIRA is indicated for the treatment of severe chronic plaque psoriasis in children and adolescents from 4 years of age who have had an inadequate response to or are inappropriate candidates for topical therapy and phototherapies.
Some patients may not be suitable for HUMIRA. You are strongly advised to read the prescribing information, which can be found at the top of this webpage and below, and the Summary of Product Characteristics (SmPC) which are available online in the Electronic Medicines Compendium (EMC).
HUMIRA 40 mg solution for injection in pre-filled pen CLICK HERE.
ADALIMUMAB PRESCRIBING INFORMATION CLICK HERE.
References
1. SKYRIZI Summary of Product Characteristics
2. HUMIRA Summary of Product Characteristics