UK-RISN-230338. Date of preparation January 2024.
Dosing
Gain a clear understanding of the straightforward dosing schedule and administration process for SKYRIZI.
Featured content
The York Experience
Dr Keith Wu and Pauline Stopford- Taylor revisit their experience setting up a specialist service in York. They also illustrate the convenience in dosing and impact of SKYRIZI by sharing their treatment journey with a patient whose psoriasis of the hands was affecting his ability to act as a primary carer for his wife.
UK-RISN-230323. Date of preparation February 2024.
Home delivery and tailored nurse services offers simple dosing and support
- Homecare registration, delivery and injection administration providing care to your patients in their home.
- Support that adapts to the evolving needs of your patients. With AbbVie Care patients benefit from tailored face-to-face and telephone support* from Homecare nurses. Ensuring the continuation of care from the hospital to their home.
- Following titration one dose per delivery, minimising the risk of waste due to potential treatment changes and reducing the burden on patients by alleviating the need to store multiple doses at their own home.
- Clinical evaluations provided by the SKYRIZI Homecare nurse so that you can feel confident knowing that your patients are supported between hospital visits.
*Telephone support not offered in Scotland.
AbbVie Care is developed and funded by AbbVie Ltd.
For all patients enrolled in the homecare support program provided through AbbVie Care.
References
- Reich K, et al. Lancet 2019; 394: 576-586.
- Gordon KB, et al. Lancet 2018; 392: 650-661.
- Warren RB, et al. Risankizumab vs Secukinumab in Patients with Moderate-to-Severe Plaque Psoriasis: A Phase 3 Trial, Presented at AAD 2020.
- SKYRIZI: Summary of Product Characteristics.
UK-RISN-230322. Date of preparation: February 2024.
Important safety information for SKYRIZI®▼(risankizumab) in Psoriasis and Psoriatic Arthritis¹
SKYRIZI should be initiated and supervised by healthcare professionals experienced in the diagnosis and treatment of Psoriasis and Psoriatic arthritis.
Some patients may not be suitable for SKYRIZI (risankizumab). You are strongly advised to read the prescribing information, which can be found at the top of this webpage and below, and the Summary of Product Characteristics (SmPC) which are available online in the Electronic Medicines Compendium (EMC) from the links below.
SKYRIZI 150 mg solution for injection in pre-filled pen CLICK HERE.
SKYRIZI 150 mg solution for injection in pre-filled syringe CLICK HERE.
SKYRIZI PRESCRIBING INFORMATION CLICK HERE.
SKYRIZI is contraindicated in patients;
- with hypersensitivity to the active substance or to any of the excipients,
- with clinically important active infections (e.g. active tuberculosis).
Cautions (See Prescribing Information and SmPC for full details including screening and monitoring requirements):
It is preferable to avoid the use of SKYRIZI during pregnancy. Women of childbearing potential should use an effective method of contraception during treatment and for at least 21 weeks after treatment. It is unknown whether risankizumab is excreted in human milk. A decision should be made whether to discontinue/abstain from risankizumab therapy, taking into account the benefit of breast-feeding to the child and the benefit of risankizumab therapy to the woman.
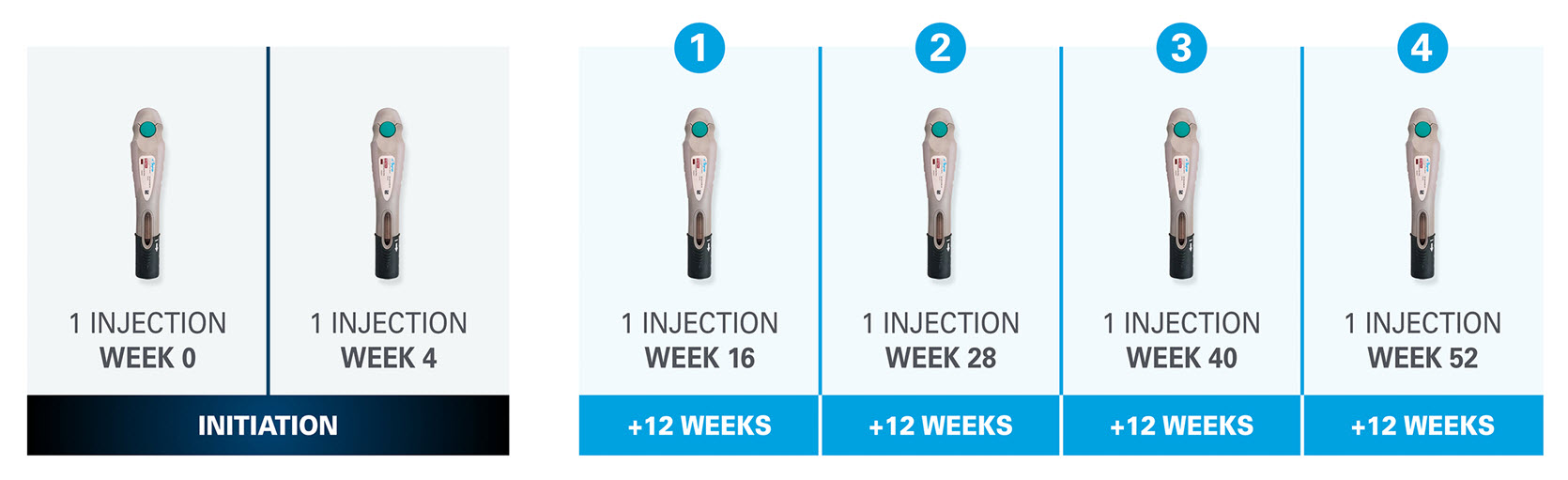
Dosing in Psoriasis and Psoriatic Arthritis
The recommended dose of SKYRIZI is 150mg administered by subcutaneous injection at Week 0, Week 4, and every 12 weeks thereafter.
Adverse reactions
For adverse reactions, please refer to the prescribing information and the SKYRIZI summary of product characteristics available online in the Electronic Medicines Compendium via the links above.
Important safety information for HUMIRA® (adalimumab) in Psoriasis and Psoriatic Arthritis²
HUMIRA is intended for use under the guidance and supervision of a physician experienced in the diagnosis and treatment of conditions for which HUMIRA is indicated.
Psoriatic arthritis: HUMIRA is indicated for the treatment of active and progressive psoriatic arthritis in adults when the response to previous disease-modifying antirheumatic drug therapy has been inadequate. HUMIRA has been shown to reduce the rate of progression of peripheral joint damage as measured by X-ray in patients with polyarticular symmetrical subtypes of the disease and to improve physical function.
Psoriasis: HUMIRA is indicated for the treatment of moderate-to-severe chronic plaque psoriasis in adult patients who are candidates for systematic therapy
Paediatric plaque psoriasis: HUMIRA is indicated for the treatment of severe chronic plaque psoriasis in children and adolescents from 4 years of age who have had an inadequate response to or are inappropriate candidates for topical therapy and phototherapies.
Some patients may not be suitable for HUMIRA. You are strongly advised to read the prescribing information, which can be found at the top of this webpage and below, and the Summary of Product Characteristics (SmPC) which are available online in the Electronic Medicines Compendium (EMC).
HUMIRA 40 mg solution for injection in pre-filled pen CLICK HERE.
ADALIMUMAB PRESCRIBING INFORMATION CLICK HERE.
References
1. SKYRIZI Summary of Product Characteristics
2. HUMIRA Summary of Product Characteristics





.jpg)